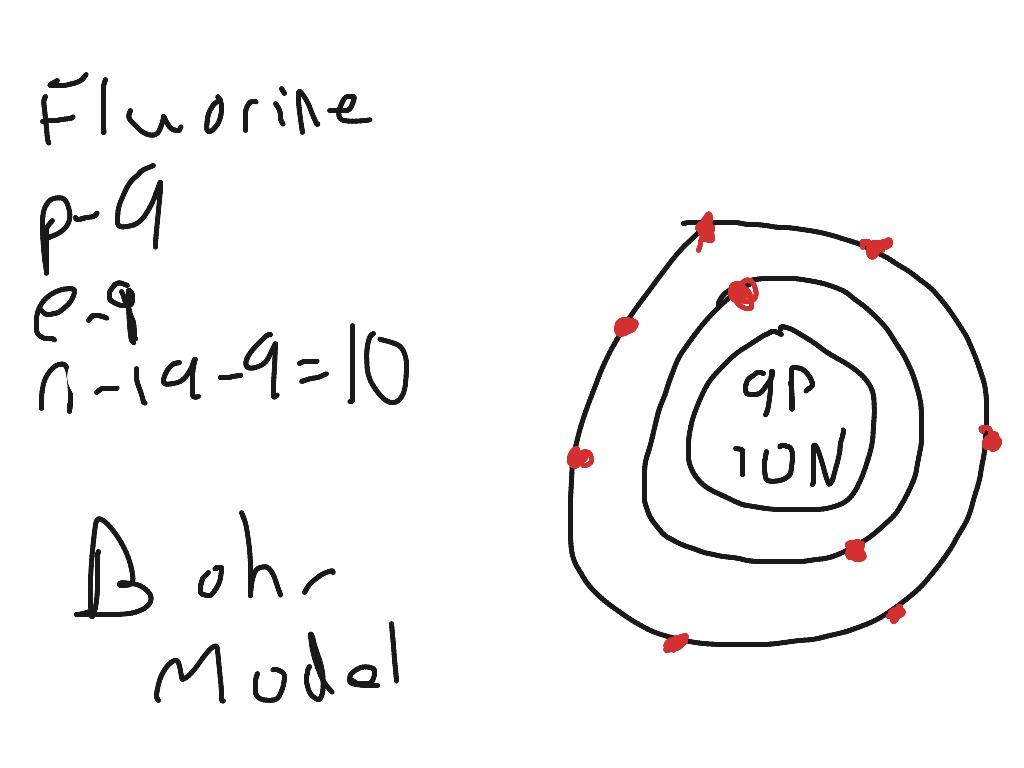
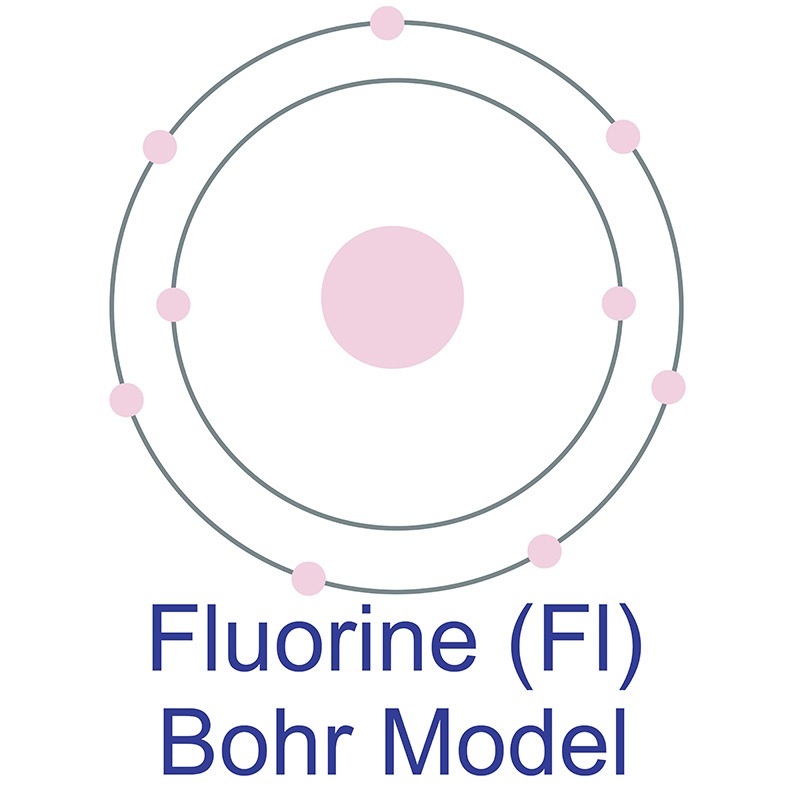
Fluorine has 2 electrons in its first shell and 7 in its second.Check me out: http://www.chemistnate.com Physical & Theoretical Chemistry Supplemental Modules (Physical and Theoretical Chemistry) Electronic Structure of Atoms and Molecules
Fluorine Bohr model Science ShowMe
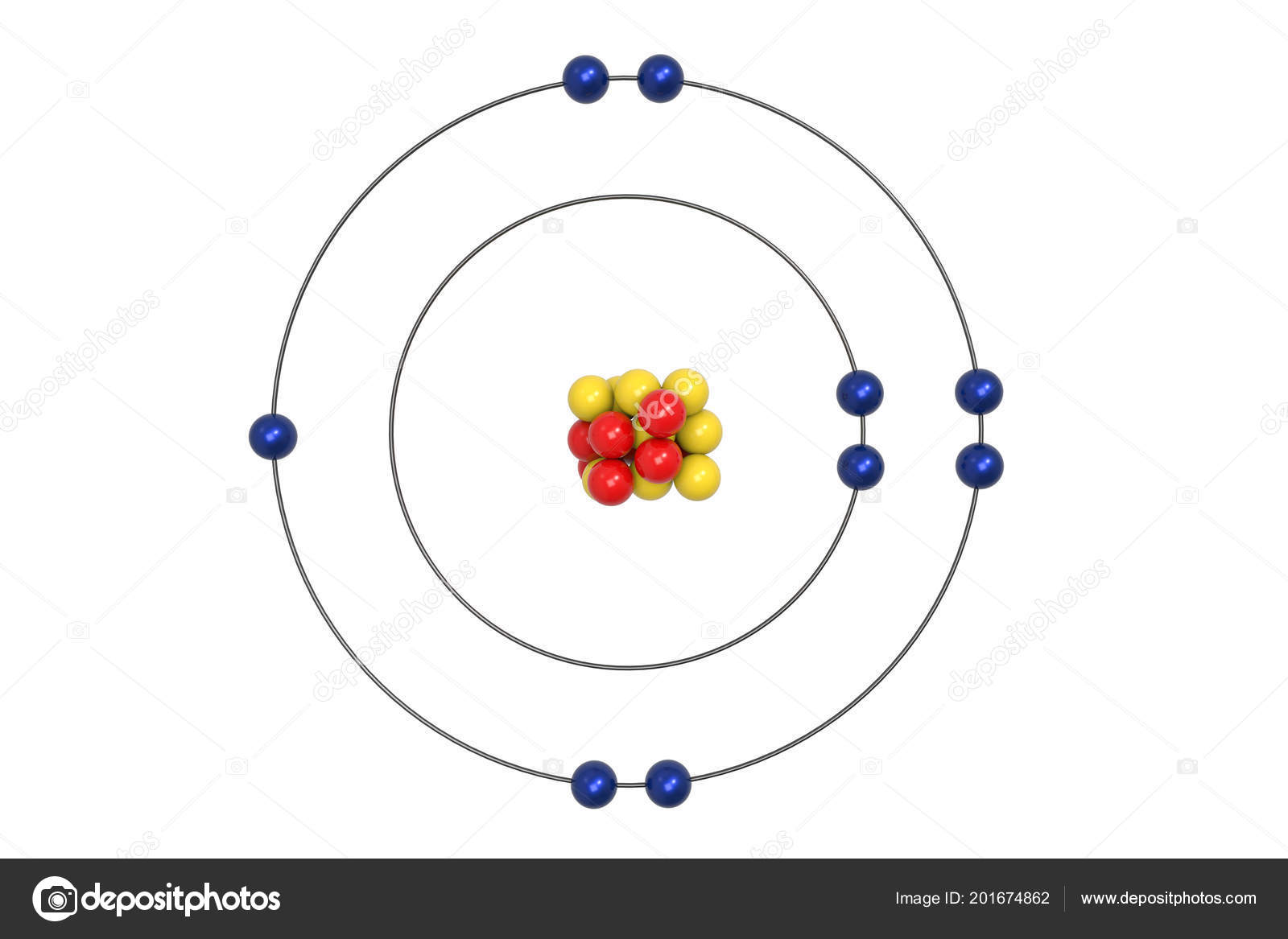
Bohr diagram is very interesting and easy to draw. Here, we will draw the Bohr diagram of the Fluorine atom with some simple steps. Steps to draw the Bohr Model of Fluorine atom 1. Find the number of protons, electrons, and neutrons in the Fluorine atom This video expectation is to understand how the Bohr-Rutherford was designed and effectively use a template to draw the Bohr-Rutherford diagram of the first. In this video we'll look at the atomic structure and Bohr model for the Fluorine atom (F). We'll use a Bohr diagram to visually represent where the electrons. This is the Bohr-Rutherford Diagram for Fluorine (Atomic Number 9). Fluorine is the ninth element of the Periodic Table. As shown, Fluorine has nine protons (i.e., 9p) and ten neutrons (i.e., 10n). Thus, its Atomic Mass is 19. Fluorine also has nine total electrons -- two electrons in Orbit 1, seven electrons in Orbit 2.
Fluorine Atom Bohr Model Proton Neutron Electron Illustration Stock
This page contains materials for the session on the atomic models of Rutherford and Bohr. It features a 1-hour lecture video, and also presents the prerequisites, learning objectives, reading assignment, lecture slides, homework with solutions, and resources for further study. More. Embed this widget ». Added Aug 1, 2010 by JB1295 in Chemistry. Gives the Lewis Dot structure for any element. Send feedback | Visit Wolfram|Alpha. Lewis Dot Diagram of. Submit. Get the free "Bohr Model Widget" widget for your website, blog, Wordpress, Blogger, or iGoogle. The Bohr model is a relatively primitive model of the hydrogen atom, compared to the valence shell model. As a theory, it can be derived as a first-order approximation of the hydrogen atom using the broader and much more accurate quantum mechanics and thus may be considered to be an obsolete scientific theory. Bohr Diagram: The First Element In order to make a Bohr diagram, you need to know the number of protons, neutrons, and electrons the element has. In this section, we'll show a sample Bohr diagram for hydrogen. H —Hydrogen 1 proton 1 electron 0 neutrons
Fluorine Bohr Model
Bohr's model of the atom can be combined with Rutherford's model in diagrams that summarize the numbers and positions of all three subatomic particles. For example, consider the following diagram for Phosphorous: There are certain rules to follow when drawing these diagrams: A circle is drawn in the center to represent the nucleus of the atom. The simplest example of the Bohr Model is for the hydrogen atom (Z = 1) or for a hydrogen-like ion (Z > 1), in which a negatively charged electron orbits a small positively charged nucleus. Electromagnetic energy will be absorbed or emitted if an electron moves from one orbit to another. Only certain electron orbits are permitted.
1 Ry = e4me 8ϵ2 0h2 = 2.18 × 10 − 18 J. and this simplifies the allowed energies predicted by the Bohr model (Equation 7.4.11) as. En = − (2.18 × 10 − 18)Z2 n2 J = − Z2 n2 Ry. Hence, the energy of the electron in an atom also is quantized. Equation 7.4.12 gives the energies of the electronic states of the hydrogen atom. Schematic diagram of the Rutherford-Bohr atomic model with one orbital electron of mass m e and a finite nucleus of mass M. Both the orbital electron and the nucleus revolve about their common center-of-mass. Full size image. The following relationships will be useful in our calculations.

Grade 9 BohrRutherford Diagram Fluorine YouTube
In 1913 Niels Bohr combined Rutherford's concept of the nuclear atom with Planck's idea of the quantized nature of the radiative process and developed, from first principles, an atomic model that successfully deals with one-electron structures like the hydrogen atom and one-electron ions such as singly ionized helium, doubly ionized lithium, etc. forming a hydrogen-like or hydrogenic. Set up the diagram. To set up the diagram, you will need a circle in the middle. This will represent the nucleus. Here you will write the number of protons and neutrons as shown below in this example using sodium (Na) Add in orbitals and electrons. In the last step you will need to draw circles around the nucleus.




