460 Share 99K views 10 years ago A step-by-step explanation of how to draw the CH3Cl Lewis Dot Structure (Chloromethane). For the CH3Cl structure use the periodic table to find the total. Contents show Lewis Structure of Chloromethane (CH3Cl) The Lewis structure is a pictorial representation showing the electrons present in the valence shell in an atom. The diagram is drawn to determine how the valence electrons of different atoms participate in the bond formation to form a molecule.

Ch3cl lewis structure, Characteristics13 Must To Know Facts Lambda Geeks
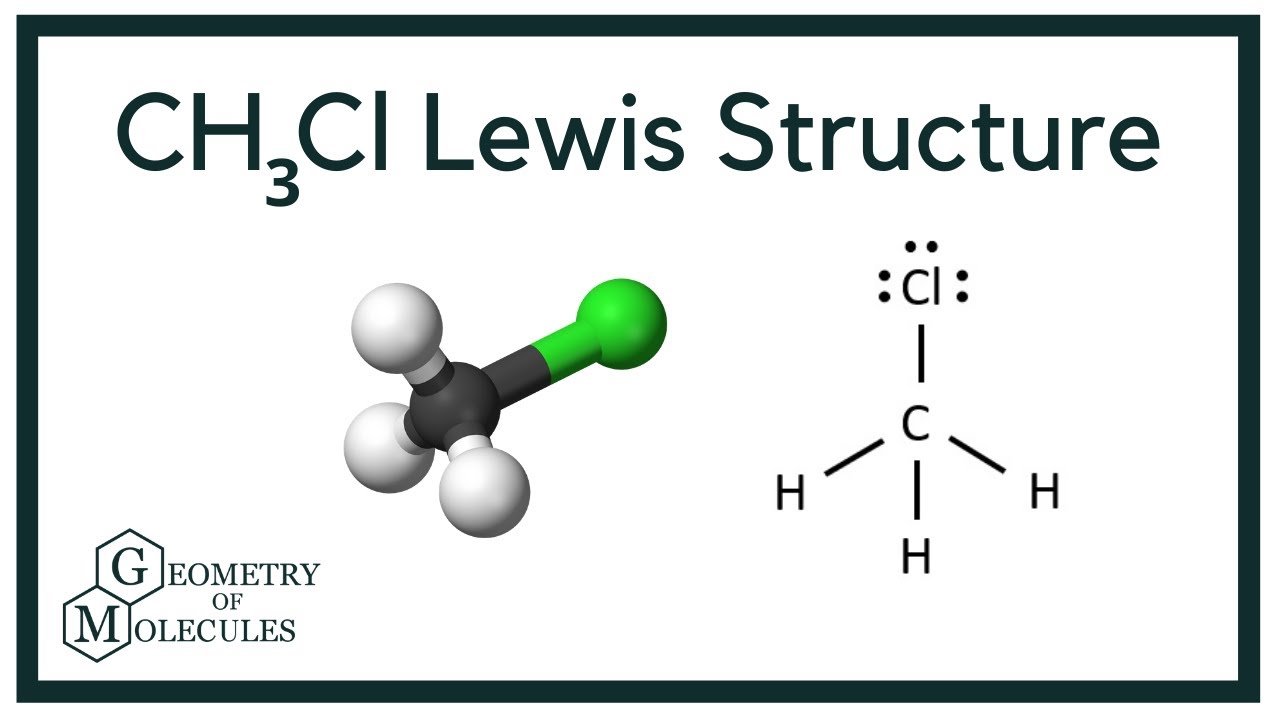
In Lewis structure, we use dots to represent electrons and lines to show bonds formed between two atoms. Here we have three types of atoms in CH3Cl: Carbon, Hydrogen, and Chlorine. Out of all these atoms, Carbon is the least electronegative one, and hence we will place it in the central position. Chlorine is the most electronegative atom. Added Jun 9, 2014 by WebTester in Chemistry This widget gets the Lewis structure of chemical compounds. Send feedback | Visit Wolfram|Alpha Get the free "Lewis Structure Finder" widget for your website, blog, Wordpress, Blogger, or iGoogle. Find more Chemistry widgets in Wolfram|Alpha. In the CH 3 Cl Lewis structure, there are four single bonds around the carbon atom, with three hydrogen atoms and one chlorine atom attached to it, and on the chlorine atom, there are three lone pairs. CH3Cl Lewis Structure - How to Draw the Lewis Structure for CH3Cl (Chloromethane) Watch on Contents Steps #1 First draw a rough sketch Hey Guys!In this video, we are going to learn about the Lewis dot structure of Chloromethane or Methyl chloride having a chemical formula of CH3Cl. The molec.
What Is Ch3cl Lewis Structure?
The first step is to sketch the Lewis structure of the CH3Cl molecule, to add valence electron around the carbon atom; the second step is to add valence electrons to the one chlorine and three hydrogen atoms, and the final step is to combine the step1 and step2 to get the CH3Cl Lewis Structure. Chloromethane (CH 3 Cl) Lewis Structure Chloromethane (CH 3 Cl) contains one carbon atom, three hydrogen atoms and one chlorine atom. In the lewis structure of CH 3 Cl, carbon atom is located as the center atom and other atoms have made bonds with carbon atom. CH 3 Cl lewis structure There are three hydrogen atoms around center carbon atom. CH3Cl lewis structure has a Carbon atom (C) at the center which is surrounded by three Hydrogen atoms (H) and one Chlorine atom (Cl). There is a single bond between the Carbon (C) & Chlorine (Cl) atom as well as between the Carbon (C) and Hydrogen (H) atoms. There are 3 lone pairs on the Chlorine atom (Cl). About this video -Lewis dot structure of CH3Cl, known as methyl chloride or chloromethane.Happy Reading :)

CHCl3 Lewis Structure, Geometry, Hybridization, and Polarity
The Lewis structure of CH3Cl consists of a carbon (C) atom at the center which is bonded to three hydrogens (H) atoms and one atom of chlorine (Cl). There are a total of 4 electron density regions around the central C atom in the CH3Cl Lewis structure. 3.1: Lewis Structures. Chemical bond refers to the forces holding atoms together to form molecules and solids. This force is of an electric nature, and the attraction between electrons of one atom to the nucleus of another atom contributes to what is known as chemical bonds.
1.Determine the number of lone pairs on the terminal hydrogen atom of the CH3Cl Lewis structure. we need to calculate out how many there are on the terminal hydrogen atom of the Lewis structure. As we know clearly, the hydrogen atom has only s orbital in the ground state. It can take a maximum of two electrons. Lewis structure of CH3Cl contains a single bond between the Carbon (C) & Hydrogen (H) atoms as well as between the Carbon (C) & Chlorine (Cl) atom. The Carbon atom (C) is at the center and it is surrounded by three Hydrogen (H) and one Chlorine atom (Cl). The Chlorine atom has 3 lone pairs.
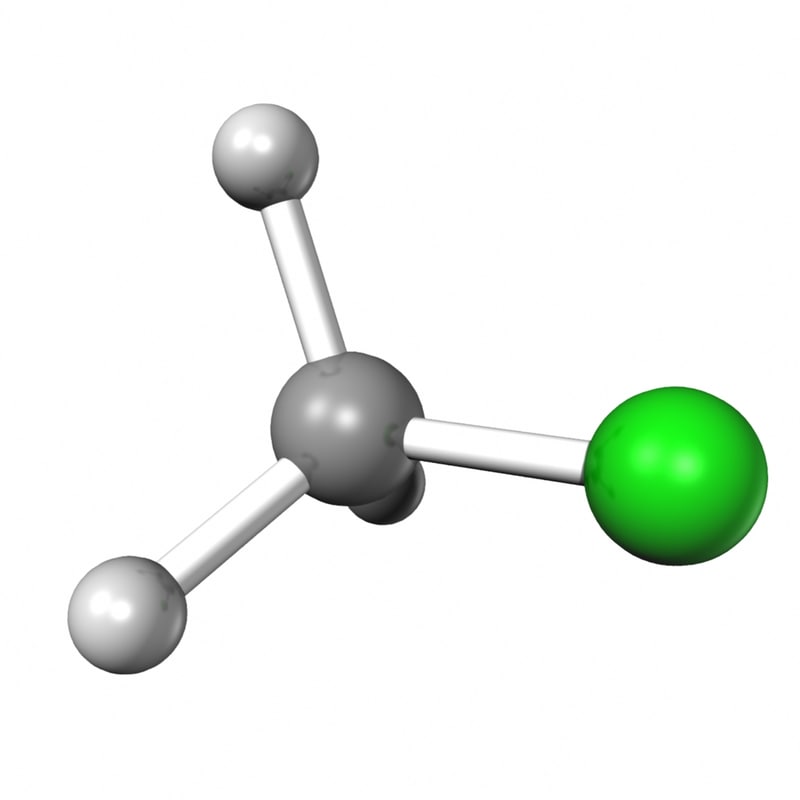
ch3cl 3d obj
According to this model, valence electrons in the Lewis structure form groups, which may consist of a single bond, a double bond, a triple bond, a lone pair of electrons, or even a single unpaired electron, which in the VSEPR model is counted as a lone pair. Because electrons repel each other electrostatically, the most stable arrangement of electron groups (i.e., the one with the lowest. To determine if CH 3 Cl is polar or nonpolar, we need to first determine its geometry. This presumes knowing the rules for drawing a correct Lewis structure and you can find more details about Lewis structures here. CH 3 Cl has 4 + 3 + 7 = 14 valence electrons. Carbon goes in the middle and is bonded to three hydrogen and one chlorine atom.




