In order to write the Na electron configuration we first need to know the number of electrons for the Na atom (there are 11 electrons). When we write the configuration we'll put all 11 electrons in orbitals around the nucleus of the Sodium atom. In writing the electron configuration for sodium the first two electrons will go in the 1s orbital. Because lithium's final electron goes into the 2s subshell, we write the electron configuration of a lithium atom as 1s 2 2s 1. The shell diagram for a lithium atom is shown below. The shell closest to the nucleus (first shell) has 2 dots representing the 2 electrons in 1 s , while the outermost shell ( 2 s ) has 1 electron.

FileElectron shell 011 sodium.png Wikimedia Commons
The arrangement of electrons in sodium in specific rules in different orbits and orbitals is called the electron configuration of sodium. The electron configuration of sodium is [ Ne] 3s 1 , if the electron arrangement is through orbitals. Electron configuration can be done in two ways. Electron configuration through orbit (Bohr principle) The specific arrangement of electrons in orbitals of an atom determines many of the chemical properties of that atom. Orbital Energies and Atomic Structure.. Sodium cation loses one electron, so Na +: 1s 2 2s 2 2p 6 3s 1 = Na +: 1s 2 2s 2 2p 6. P: 1s 2 2s 2 2p 6 3s 2 3p 3. Electron configuration The arrangements of electrons above the last (closed shell) noble gas. Melting point The temperature at which the solid-liquid phase change occurs.. Sodium is essential to all living things, and humans have known this since prehistoric times. Our bodies contain about 100 grams, but we are constantly losing sodium in. The electron configuration of sodium is \(1s^2 2s^2 2p^6 3s^1\) (Table \(\PageIndex{1}\)). The first ten electrons of the sodium atom are the inner-shell electrons and the configuration of just those ten electrons is exactly the same as the configuration of the element neon \(\left( Z=10 \right)\).. An orbital diagram is the more visual way.
Electron arrangements
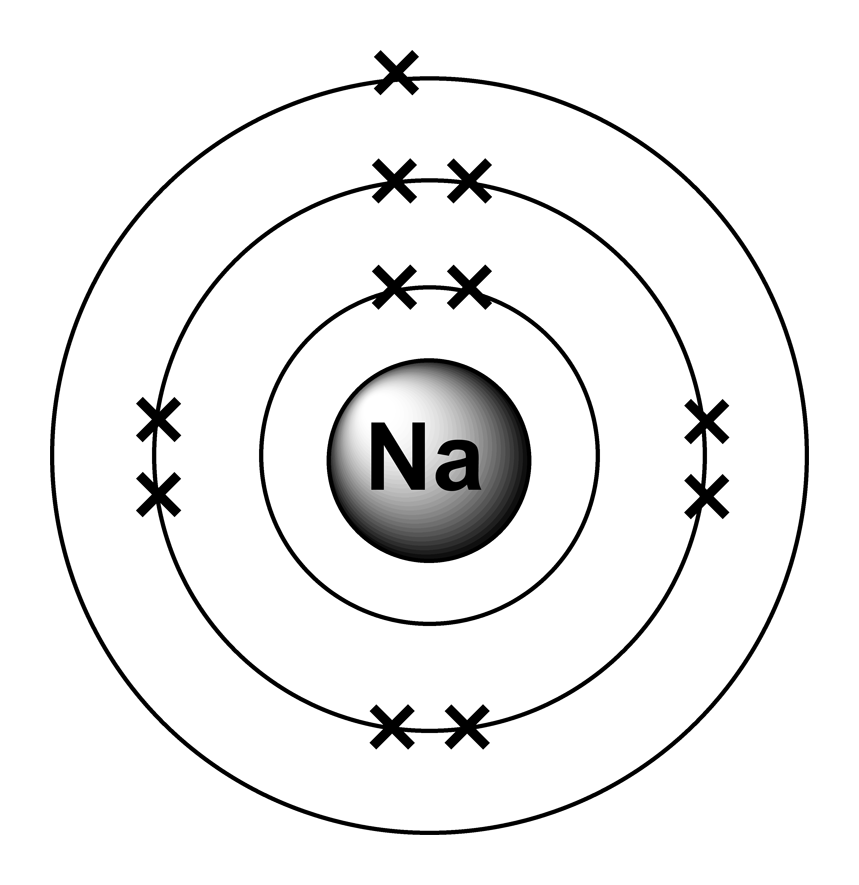
1 to 20: Predicting an electron arrangement The electron arrangement of an atom can be predicted from its atomic number. For example, the atomic number of sodium is 11. Sodium atoms. If we look at the arrangement of electrons in a sodium atom, we will find that two electrons fill the first shell, eight electrons fill the second shell, and there's one electron left to go in the third shell. But the question remains. The electron arrangement of an element is related to its position on the periodic table. The electron arrangement of sodium (2.8.1) shows that sodium, Na: is in period 3 is in group 1. Sodium (Na) is the first element in the 3rd row (Period 3) in the periodic table. This means that the first shell and second shells of Na atom are filled to the maximum number of electrons. The first shell (1s) is filled with 2 electrons. The second shell (2s and 2p) has a total of 8 electrons. And, the third (last) shell has 1 electron.

Atom Sodium Model Stock Illustration Download Image Now iStock
A step-by-step description of how to write the electron configuration for Sodium (Na). In order to write the Na electron configuration we first need to know. The arrangement of electrons in the orbitals of an atom is called the electron configuration of the atom. We describe an electron configuration with a symbol that contains three pieces of information ( Figure 6.25 ): The number of the principal quantum shell, n,
In this video we will write the electron configuration for Na+, the Sodium ion. We'll also look at why Sodium forms a 1+ ion and how the electron configurati. The electron arrangement of the sodium ion is now the same as that of the noble gas neon. Consider a similar process with magnesium and aluminum. In this case, the magnesium atom loses its two valence electrons in order to achieve the same arrangement as the noble gas neon and a charge of \(2+\). The aluminum atom loses its three valence.

Sodium Atom Science Notes and Projects
The electron configuration of a neutral sodium atom is 1s^2 2s^2 2p^6 3s^1. In this configuration we note that there is only one electron in the 3rd energy level. Atoms prefer to gain the stability of octet, by having eight electrons in the outer shell, the electrons of the s and p orbitals. . They begin to occupy the next shell only when this shell becomes full. For elements with atomic number 1 to 20: Predicting an electron arrangement The electron arrangement of an atom can be.




