The electron configuration for Gallium, Ga is 1s^2 2s^2 2p^6 3s^2 3p^6 3d^10 4s^2 4p^1 Gallium, Ga has 31 protons and 31 electrons. The superscripts represent the electrons present in each region of the periodic table. The sum of these superscripts should equal the atomic number for a neutral atom. The arrangement of electrons in gallium in specific rules in different orbits and orbitals is called the electron configuration of gallium. The electron configuration of gallium is [ Ar] 3d 10 4s 2 4p 1 , if the electron arrangement is through orbitals. Electron configuration can be done in two ways.

Electron Configuration for Gallium (Ga, Ga3+ ion)
267 38K views 3 years ago A step-by-step description of how to write the electron configuration for Gallium (Ga). In order to write the Ga electron configuration we first need to know. Element Gallium (Ga), Group 13, Atomic Number 31, p-block, Mass 69.723. Sources, facts, uses, scarcity (SRI), podcasts, alchemical symbols, videos and images.. Electron configuration The arrangements of electrons above the last (closed shell) noble gas. Melting point Gallium is a chemical element; it has symbol Ga and atomic number 31.. [Xe]4f 14 5d 10 6s 2 electron configuration, which is liquid at room temperature. The 3d 10 electrons do not shield the outer electrons very well from the nucleus and hence the first ionisation energy of gallium is greater than that of aluminium. Referring to either Figure 2.6.3 2.6. 3 or 2.6.4 2.6. 4, we would expect to find the electron in the 1 s orbital. By convention, the ms = +1 2 m s = + 1 2 value is usually filled first. The electron configuration and the orbital diagram are: Following hydrogen is the noble gas helium, which has an atomic number of 2.

Ga electronic configurationHow to write electronic configuration of
The gallium atom requires five more valence electrons in its p-orbital to attain noble gas configuration. What is the complete electron configuration of gallium? The complete electron configuration of gallium is 1s2 2s 2 2p6 3s2 3p6 4s2 3d10 4p1. Gallium is the chemical element with the atomic number 31 and symbol Ga on the periodic table. It is in the Boron family (group 13) and in period 4. Gallium was discovered in 1875 by Paul Emile Lecoq de Boisbaudran. Boisbaudran named his newly discovered element after himself, deriving from the Latin word, "Gallia," which means "Gaul.". Full electron configuration of gallium: 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p1 zinc ← gallium → germanium Gallium, complete electron configuration. 1). You can effortlessly find every single detail about the elements from this single Interactive Periodic table. 2). You will get the detailed information about the periodic table which will convert a newbie into pro. 3). You will also get the HD images of the Periodic table (for FREE).

Gallium (Ga). Diagram of the nuclear composition and electron
In the above electron configuration, the highest energy level (4) is marked with green color. The 4 th energy level contains 4s and 4p subshells. There are 2 electrons in the 4s subshell and 1 electron in the 4p subshell. So gallium has a total of 2 + 1 = 3 valence electrons. Your feedback matters. Gallium-67 (half-life 3.3 days) is a gamma-emitting isotope (the gamma emitted immediately after electron-capture) used in standard nuclear medical imaging, in procedures usually referred to as gallium scans. Stable Isotopes Typical Unstable Isotopes Electrons and Electron Configuration
Here, the electron configuration of gallium ion(Ga 3+) is 1s 2 2s 2 2p 6 3s 2 3p 6 3d 10. This gallium ion(Ga 3+) has thirty-one protons, thirty-nine neutrons, and twenty-eight electrons. Gallium ion: Protons: Neutrons: Electrons: Ga 3+ 31: 39: 28: Number of protons, neutrons and electrons for the gallium ion(Ga 3+) Electronic configuration of Gallium (Ga): Gallium is a p-block element having atomic number 31. The group number and period number of Gallium are 13 and 4 respectively. Therefore, the electronic configuration of Ga is [ Ar] 3 d 10 4 s 2 4 p 1. Suggest Corrections.
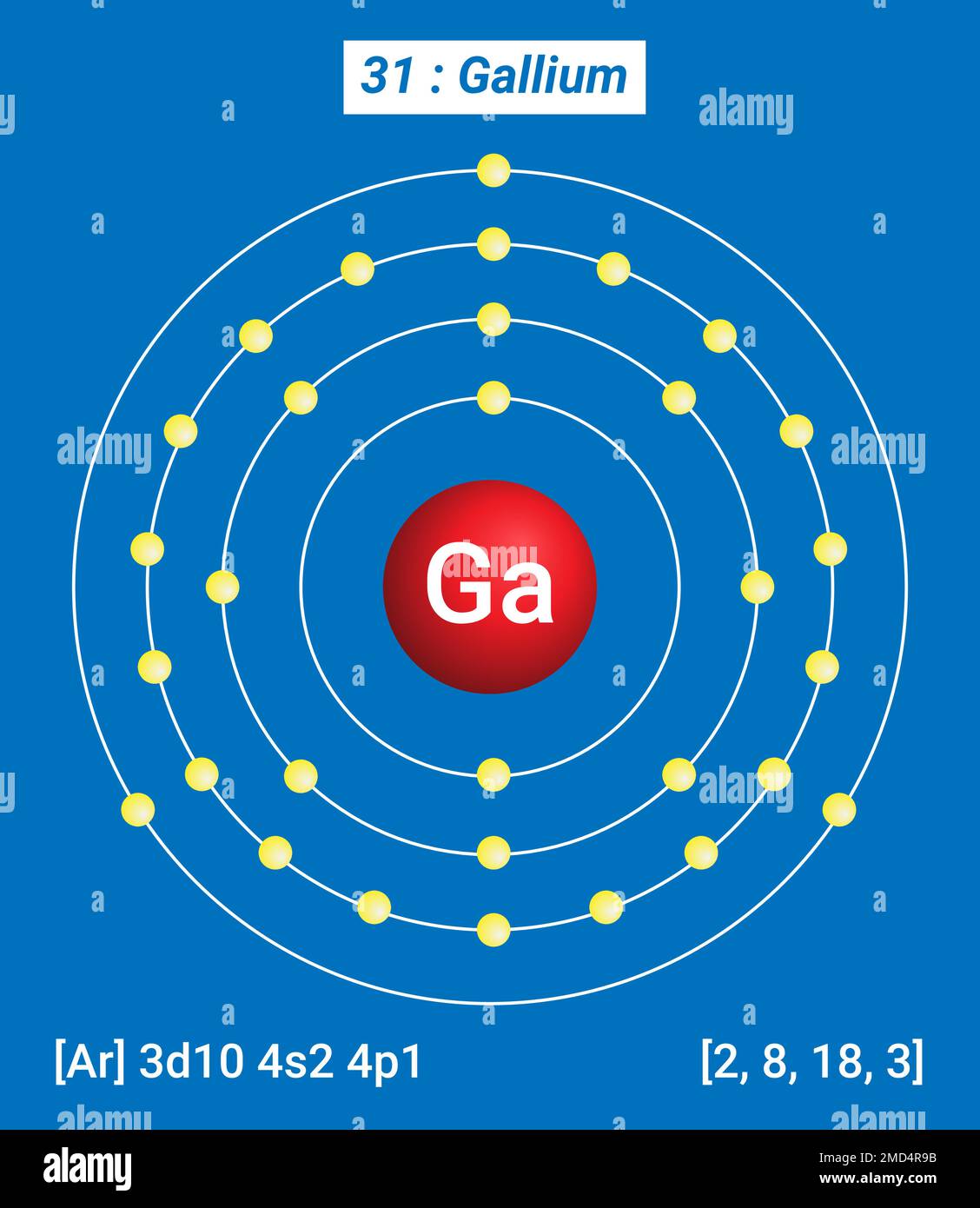
Ga Gallium, Periodic Table of the Elements, Shell Structure of Gallium
#1 Using aufbau principle #2 Using periodic table #3 From its Bohr model #4 From its orbital diagram Let's break down each method in detail. Using aufbau principle First, find electrons of gallium atom Periodic table The atomic number of gallium represents the total number of electrons of gallium. Step 1: Writing the electron shell number Gallium has 4 electron shells which are written as 1, 2, 3, 4 Step 2: Putting orbital notations after the number of electron shells Gallium atom is consists of 3 orbitals such as 's', 'p' and 'd' Step 3: Calculating the number of electron in orbitals




