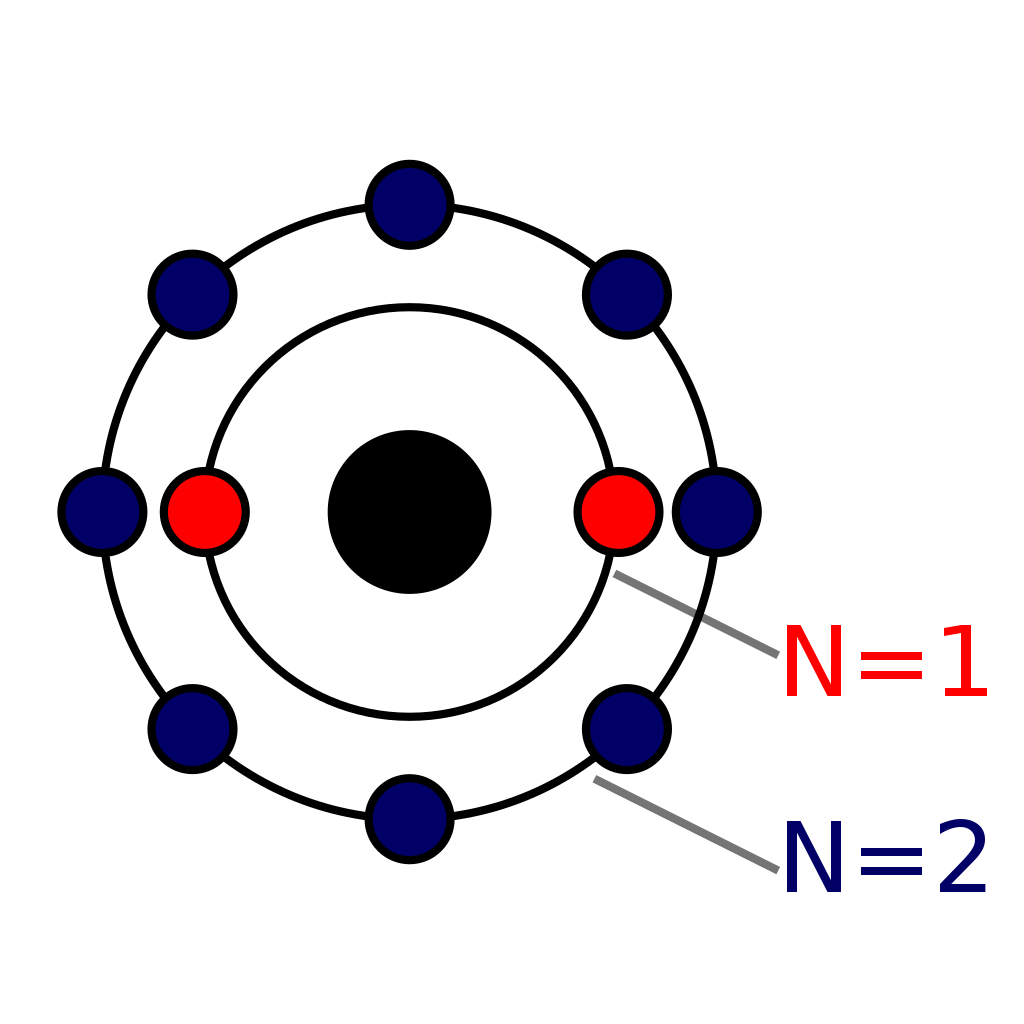
These are represented by the symbol p +. • Neutrons: These are the charge-neutral particles located inside the nucleus. These are represented using the symbol n°. • Electrons: These are the negatively charged atomic particles that revolve around the nucleus. The electrons follow a definite path while revolving around the nucleus. Neon has 2 electrons in its first shell and 8 in its secondCheck me out: http://www.chemistnate.com

Neon Bohr Model — Diagram, Steps To Draw Techiescientist
Bohr diagrams show electrons orbiting the nucleus of an atom somewhat like planets orbit around the sun. In the Bohr model, electrons are pictured as traveling in circles at different shells, depending on which element you have. Figure 2 2 contrast the Bohr diagrams for lithium, fluorine and aluminum atoms. The shell closest to the nucleus is. Atomic Structure (Bohr Model) for the Neon (Ne) Atom Wayne Breslyn 718K subscribers Join Subscribe Subscribed 35 Share 5.7K views 1 year ago In this video we'll look at the atomic structure and. In atomic physics, the Bohr model or Rutherford-Bohr model of the atom, presented by Niels Bohr and Ernest Rutherford in 1913, consists of a small, dense nucleus surrounded by orbiting electrons. The Bohr Model is a modification of an earlier atomic model, the Rutherford Model. The Bohr Model has an atom with a positively-charged nucleus surrounded by negatively-charged electrons that have circular, planetary-like orbits. Today, we know that the Bohr Model has some inaccuracies, but it's still used because of its simple approach to.
Neon Bohr Diagram
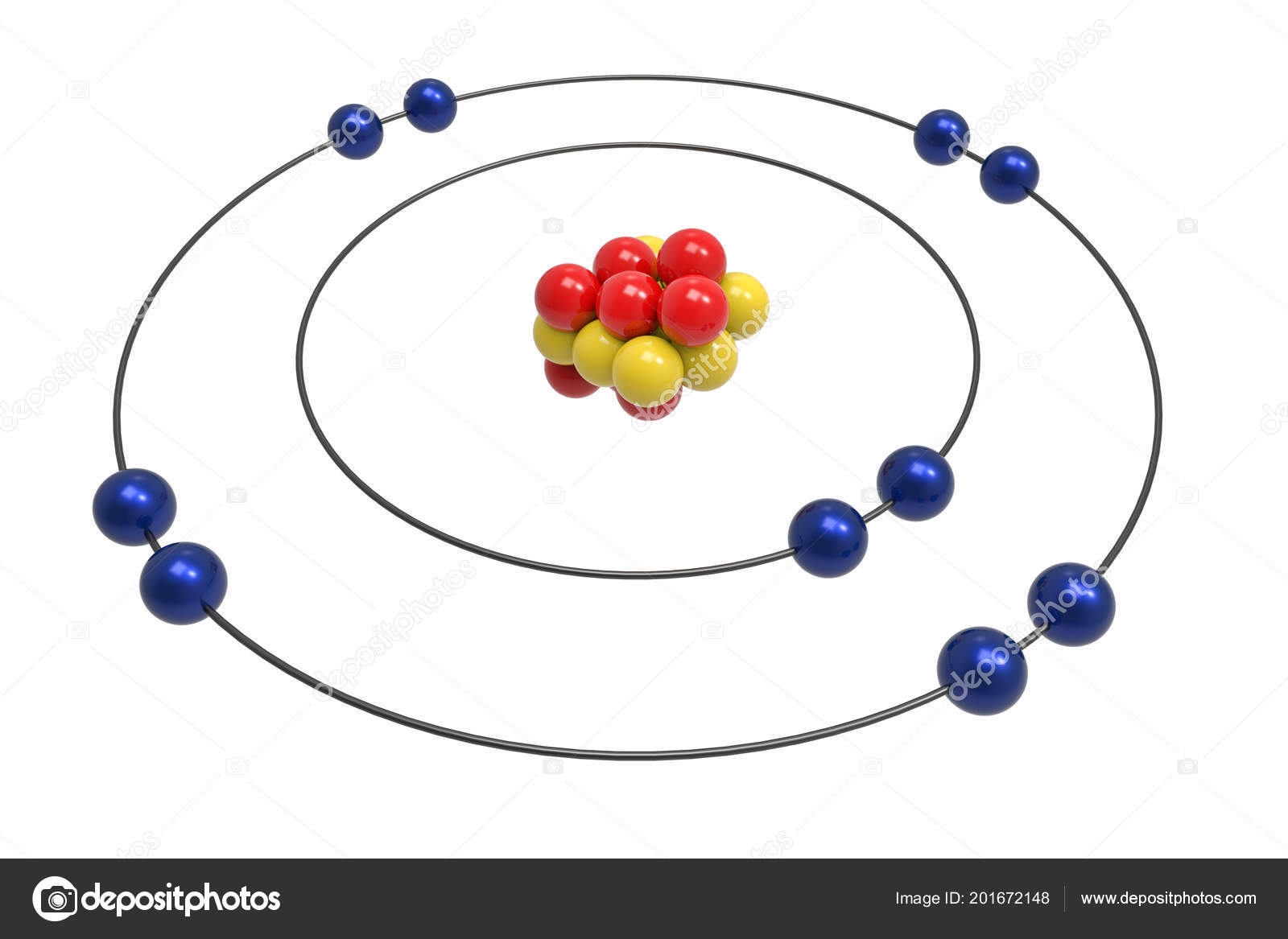
Figure 2.5. 1: Bohr diagrams: Bohr diagrams indicate how many electrons fill each principal shell. Group 18 elements (helium, neon, and argon are shown) have a full outer, or valence, shell. A full valence shell is the most stable electron configuration. Elements in other groups have partially-filled valence shells and gain or lose electrons to. Rutherford-Bohr diagram Lewis notation If an element has more than four valence electrons, they are doubled to form pairs of dots around the element. For example, neon is represented by eight valence electrons. Rutherford-Bohr diagram Lewis notation SAMPLE QUESTIONS: 1) Draw a Lewis structure for each of the following elements: Potassium The Rutherford-Bohr atomic model is a representation of the atom as a very small. Neon Atomic # 10 Atomic mass # 20 10 electrons distributed like this: - 2 on the first level - 8 on the second level RUTHERFORD- BOHR DIAGRAM . VALENCE ELECTRONS Bohr Model of Atoms. Following the work of Ernest Rutherford and his colleagues in the early twentieth century, the picture of atoms consisting of tiny dense nuclei surrounded by lighter and even tinier electrons continually moving about the nucleus was well established. This picture was called the planetary model, since it pictured the atom as a miniature "solar system" with the electrons.
Picture bohr model for neon Bohr Model Neon Atom Proton Neutron
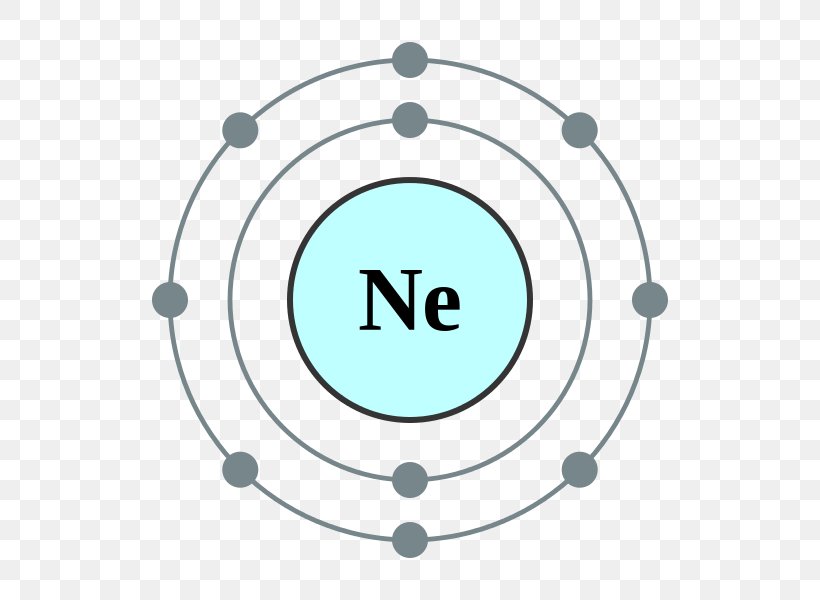
The Bohr Model of Neon (Ne) has a nucleus that contains 10 neutrons and 10 protons. This nucleus is surrounded by two-electron shells named K-shell and L-shell. The outermost shell in the Bohr diagram of Neon contains 8 electrons that also called valence electrons. Page Contents show How to draw Bohr Model of Neon (Ne)? The simplest example of the Bohr Model is for the hydrogen atom (Z = 1) or for a hydrogen-like ion (Z > 1), in which a negatively charged electron orbits a small positively charged nucleus. Electromagnetic energy will be absorbed or emitted if an electron moves from one orbit to another. Only certain electron orbits are permitted.
Bohr model: energy levels. What is the ( i ) kinetic energy of the electron, and the ( ii ) potential energy of the atom in the state, n = 2 ? Stuck? Use a hint. Learn for free about math, art, computer programming, economics, physics, chemistry, biology, medicine, finance, history, and more. Khan Academy is a nonprofit with the mission of. The drawbacks of the Rutherford model of the atom, including its apparent instability, would soon be addressed by his student, Dutch physicist Neils Bohr (1885 - 1962). To unlock this lesson you.
Bohr Model Neon
THE BOHR-RUTHERFORD MODEL - In the early 1900's, two European scientists, Neils Bohr and Ernst Rutherford,. Draw the Bohr-Rutherford diagram for neon. Protons = 10; Electrons = 10; Neutrons = 20 - 10 = 10 - Outside of the first orbit, electrons are usually drawn in pairs. November 1, 2023 by Deep The information on this page is fact-checked. Neon Bohr model The Bohr model of neon contains a nucleus having 10 protons and 10 neutrons in the center, and around this nucleus, there are two electron shells containing 10 electrons. Atomic Structure (Bohr Model) for the Neon (Ne) Atom Watch on Contents Steps




