A step-by-step explanation of how to draw the O3 Lewis Dot Structure (Ozone).For the O3 structure use the periodic table to find the total number of valence. A step-by-step explanation of how to draw the O3 Lewis Dot Structure (Ozone).For the O3 structure use the periodic table to find the total number of valence.

What is the Lewis dot structure for ozone? Chemistry Stack Exchange
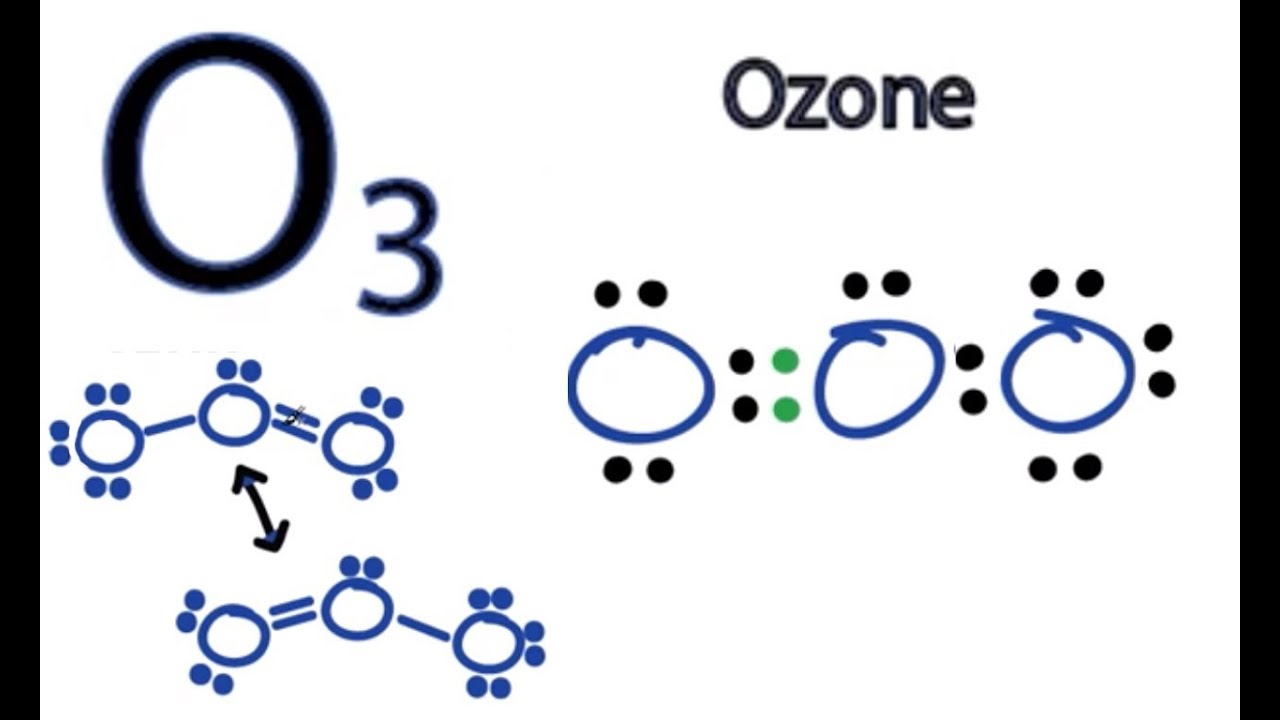
Ozone (O3) is an allotrope of oxygen and contains three oxygen atoms. In the lewis structure of ozone, there are one double bond and one single bond. Also, there are charges in two oxygen atoms in O 3 lewis structure. Lewis structure of O 3 can be drawn by starting from valence electrons of oxygen atoms in several steps. Lets start with a look at the Lewis Dot Structure of Ozone. Illustration of Resonance: Ozone. Ozone (O 3) is an allotrope of oxygen, with diatomic oxygen (O 2) being the most common form of oxygen. Ozone is a very reactive form of oxygen that has detrimental health effects (which is why ozone alerts are posted along the highways of cities), but. The ozone (O 3) molecule has two equivalent octet structures, shown below: In both cases, the Lewis dot diagram suggests that there are three kinds of oxygen atoms in the molecule, with +1, 0, and -1 formal charges. These structures also suggest that ozone should have one single and one double bond. 70 More Lewis Dot Structures. Element number 8 and a member of the Chalcogen Family or Group 16 of the periodic table. Ozone is an allotrope of oxygen, and is much less stable. Ultraviolet light cause it to decompose in our ozone layer, therefore it shielding people below it. Ozone is a pale blue gas, slightly soluble in water and much more.

Ozone Molecule Lewis Structure
Ozone (/ ˈ oʊ z oʊ n /) (or trioxygen) is an inorganic molecule with the chemical formula O 3.It is a pale blue gas with a distinctively pungent smell. It is an allotrope of oxygen that is much less stable than the diatomic allotrope O 2, breaking down in the lower atmosphere to O 2 ().Ozone is formed from dioxygen by the action of ultraviolet (UV) light and electrical discharges within the. Ozone is one of the most common examples used to study the Lewis structure. The molecule of Ozone has three oxygen atoms. It is written as O3 in the core chemistry equations. To understand the hybridization, polarity and molecular geometry of the Ozone molecule it is crucial to know the Lewis structure of the same. For starters, the Lewis dot structure for ozone does not look like that. It is not a ring. This is explained in this link: Can ozone have a triangular structure? There it is also given (one of the simpler answers): In order to form a 3-member ring your electron pairs would have to bend at 60 degrees; the energy required to push naturally. The Lewis Structure of Ozone has:* three oxygen atoms in a row. It is not a ring, although that might be tempting.* two resonance structures* a lone pair on.
O3 Lewis Structure How to Draw the Dot Structure for O3 YouTube
Sometimes one Lewis Structure is not Enough . Some molecules or ions cannot be adequately described by a single Lewis structure. For example, drawing one Lewis structure for ozone (O 3) gives us a misleading picture of the actual bonding in the molecule.If we draw a Lewis structure for O 3 (ozone), we get this:. This structure predicts that the two bonds are different lengths and strengths. Lewis Structure To be very precise, Lewis Structure is the name given to the structural representation of a molecule. It is the diagrammatic layout for understanding the nitty-gritty of chemical bonding. A very essential concept of molecular chemistry, the following steps dictate how you can successfully draw Lewis Structure: Step 1
The typical Lewis structure of ozone depicts formal charge separation Explanation: Simple VESPER requires that we distribute 3 ×6 = 18 valence electrons across 3 centres: O = O+ − O− From the left, O1, has TWO lone pairs; O2 has ONE lone pairs; and O3 has THREE lone pairs. 0:00 / 1:59 Lewis Dot Structure of O3 (Ozone) kentchemistry.com 24.7K subscribers 131K views 11 years ago I quickly take you through how to draw the Lewis Structure of O3 (Ozone). I also go.

Single Oxygen Lewis Structure
Equivalent Lewis dot structures, such as those of ozone, are called resonance structures . The position of the atoms is the same in the various resonance structures of a compound, but the position of the electrons is different. Double-headed arrows link the different resonance structures of a compound: Ozone, sometimes referred to as smog, is a gas that is formed in the atmosphere when three atoms of oxygen combine. The chemical structure of ozone is the same wherever it is found; however, there are two categories of ozone. Stratospheric Ozone is found naturally in the Earth's upper atmosphere - 6 to 30 miles above the Earth's surface.




