A step-by-step explanation of how to draw the SiH4 Lewis Dot Structure (Silicon Tetrahydride).For the SiH4 structure use the periodic table to find the total. SiH4 Lewis Structure Lewis Structure is a two-dimensional diagrammatic approach towards finding the nature of chemical bonding present inside any given molecule. Here, we use dot notations to represent the electrons, and hence this is also known as the electron-dot structure.

SiH4 Lewis Structure (Silicon Tetrahydride) YouTube
By Darshana Fendarkar In this article,"SiH4 Lewis structure", different facts like Lewis structure drawing, formal charge calculation, hybridization, structure with some detailed explanations are described below. SiH4 is also called silane or monosilane, it is a colorless flammable and poisonous gas with a strong pungent odor. An explanation of the molecular geometry for the SiH4 (Silicon Tetrahydride (Silane) including a description of the SiH4 bond angles. The electron geometry f. Chemistry learning made easy.This tutorial will help you deal with the lewis structure and moleculargeometry for silane (SiH4). Lewis structure of SiH4 contains four single bonds between each Silicon (Si) and Hydrogen (H) atoms. The Silicon atom (Si) is at the center and it is surrounded by 4 Hydrogen atoms (H). Let's draw and understand this lewis dot structure step by step. (Note: Take a pen and paper with you and try to draw this lewis structure along with me.

Silane (SiH4) molecule. Skeletal formula Stock Vector Image & Art Alamy
A step-by-step explanation of how to draw the SiH4 Lewis Dot Structure (Silicon Tetrafluoride).For the SiH4 structure use the periodic table to find the tota. Dipole Moment 9m. Octet Rule 6m. Formal Charge 2m. Lewis Dot Structures: Neutral Compounds 8m. Lewis Dot Structures: Sigma & Pi Bonds 4m. Lewis Dot Structures: Ions 9m. Lewis Dot Structures: Exceptions 6m. Lewis Dot Structures: Acids 3m. Resonance Structures 12m. SiH4 lewis structure has a Silicon atom (Si) at the center which is surrounded by four Hydrogen atoms (H). There are 4 single bonds between the Silicon atom (Si) and each Hydrogen atom (H). The Lewis structure indicates that each Cl atom has three pairs of electrons that are not used in bonding (called lone pairs) and one shared pair of electrons (written between the atoms). A dash (or line) is sometimes used to indicate a shared pair of electrons:. {SiH4}\): Si already has an octet, so nothing needs to be done.

SiH4 Molecular Geometry, Bond Angles (and Electron Geometry) YouTube
Hello everyone! Welcome back to our channel, and in today's video, we will help you do SiH4 Lewis Structure. Follow this video to know the detailed method an. So that is the SiH4 Lewis structure. This is Dr. B., and thanks for watching. Search our 100 + Lewis Structures (Opens New Window) See the Big List of Lewis Structures : Frequently Tested Lewis Structures Basic CH 4, NH 3, C 2 H 4, O 2, N 2 Intermediate O 3, BBr 3, I 3-, BrF 5, NO
Draw the Lewis structure for SiH4. Draw the molecule by placing atoms on the grid and connecting them with bonds. Include all lone pairs of electrons. Include all hydrogen atoms. To change the symbol of an atom, double-click on the atom and enter the letter of the new atom. PART B Draw the Lewis structure for CO. In the SiH 4 Lewis structure, there are four single bonds around the silicon atom, with four hydrogen atoms attached to it, and none of the atoms has a lone pair. Contents Steps #1 Draw a rough sketch of the structure #2 Next, indicate lone pairs on the atoms External links Steps To properly draw the SiH 4 Lewis structure, follow these steps:
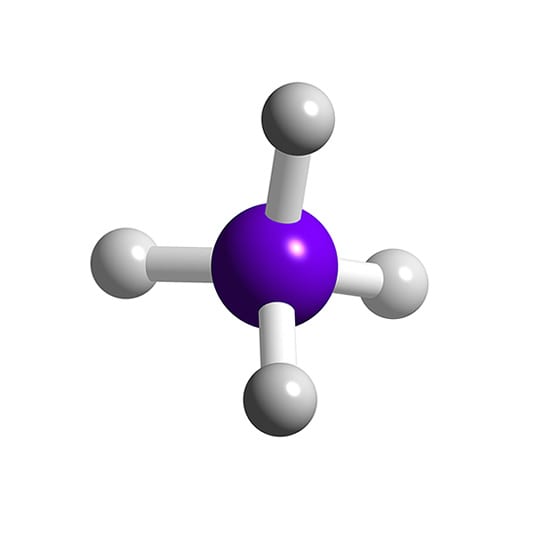
SiH4 Silane
Monosilane (SiH4) is far less well behaved than its carbon analogue methane (CH4). It is a colourless gas that is industrially relevant as a source of elemental silicon, but its pyrophoric and. SiH4 Lewis Structure - How to Draw the Lewis Structure for SiH4 (Silicon Tetrahydride) Watch on So from the above diagram we have come to know that the SiH4 molecule has four Si-H bonds. Now in the next step we have to check whether these four Si-H bonds are polar or nonpolar. And we also have to check the molecular geometry of SiH4.




