Table of Polyatomic Ions. There are a number of ions that are not individual atoms but are composed of multiple atoms that are covalently bonded together. However, this group of atoms is most stable when it has either lost of gained an electron and thus existed as a charged ion. These polyatomic ions are extremely common in chemistry and thus. A monatomic ion is an atom that has been ionized by gaining or losing electrons. The ion has a net charge because the total number of electrons is not balanced by the total number of protons in the nucleus.

Common Polyatomic Ions Names, Formulae, and Charges Compound Interest
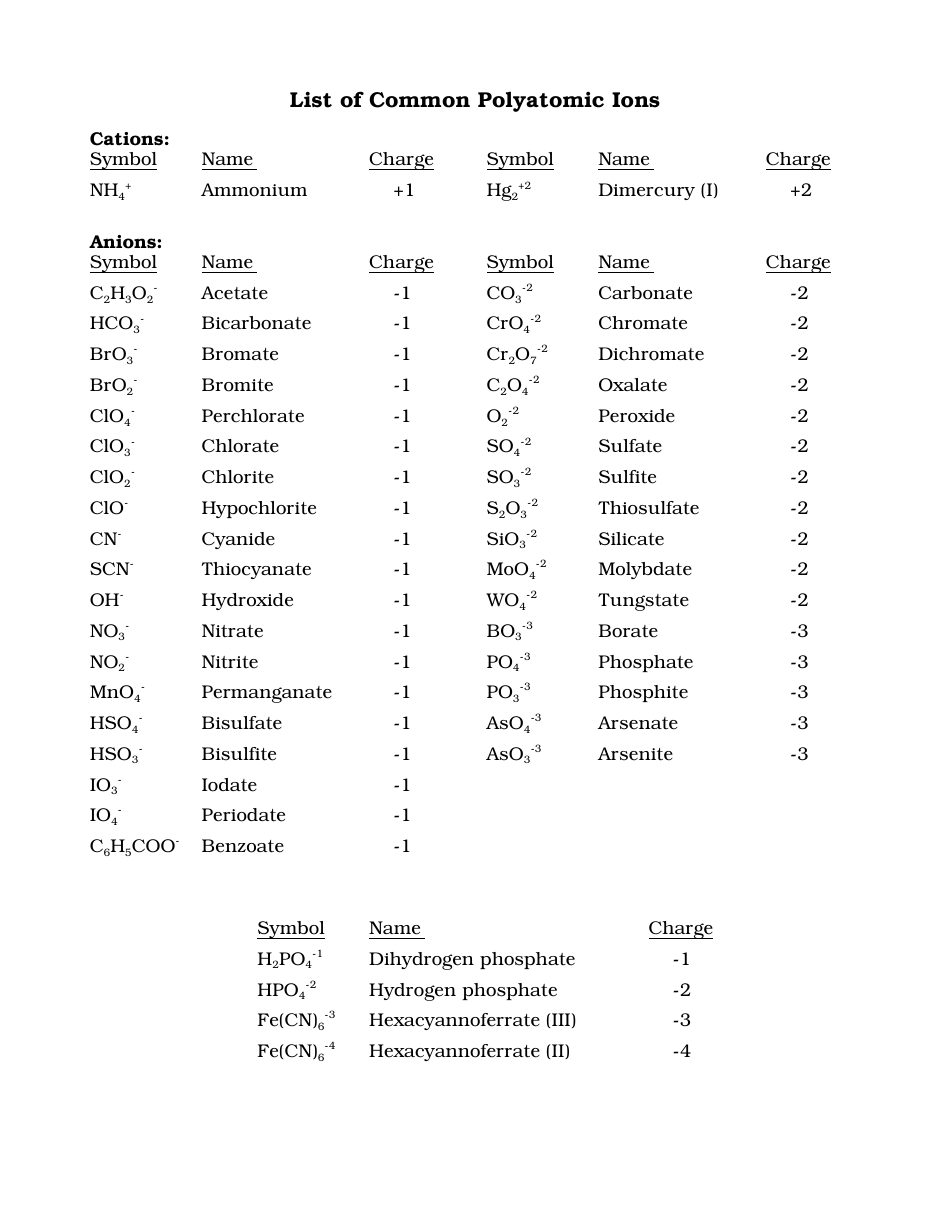
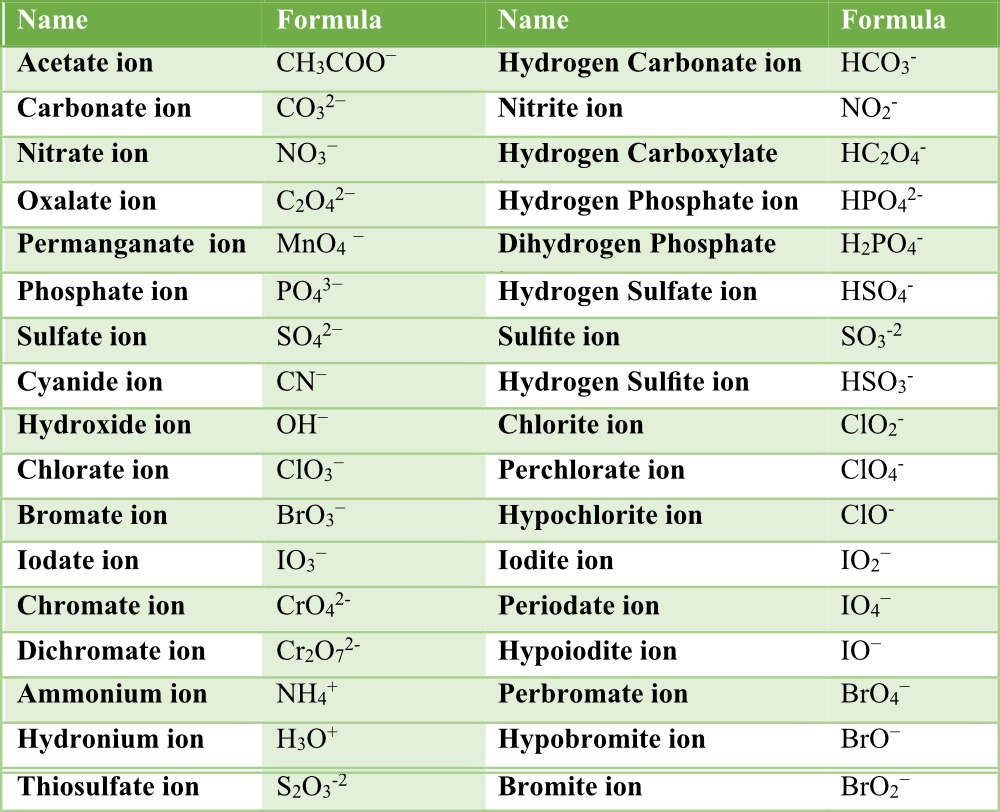
Figure 3.6. 1: Polyatomic Ions Infographic. Polyatomic ions have defined formulas, names, and charges that cannot be modified in any way. Table 3.6. 1 lists the ion names and ion formulas of the most common polyatomic ions. For example, NO 3 − is the nitrate ion; it has one nitrogen atom and three oxygen atoms and an overall −1 charge. Polyatomic ions. Polyatomic ions are molecular ions composed of two or more atoms bonded by covalent bonds and acting as a single unit, but unlike molecules, they have a net charge on them. The examples include cations like ammonium ion ( NH+4 NH 4 + ), and hydronium ion ( H3O+ H 3 O + ); and anions like hydroxide ion ( OH− OH − ), and. A polyatomic ion is an ion composed of more than one atom. The ammonium ion consists of one nitrogen atom and four hydrogen atoms. Together, they comprise a single ion with a \(1+\) charge and a formula of \(\ce{NH_4^+}\).. The table below lists a number of polyatomic ions by name and by formula. The heading for each column indicates the. POSITIVE POLYATOMIC IONS TABLE OF POLYATOMIC IONS H2PO4 - HCO3 - HC2O4 - HSO4 - HS- HSO3 - OH- ClO- IO3 - HPO4 2- NO3 - NO2 - SiO4 4- hydrogen carbonate hydrogen oxalate hydrogen sulfate hydrogen sulfide hydrogen sulfite hydroxide hypochlorite iodate nitrate nitrite orthosilicate monohydrogen phosphate dihydrogen.
Common Polyatomic Ions Chart Cations, Anions Download Printable PDF
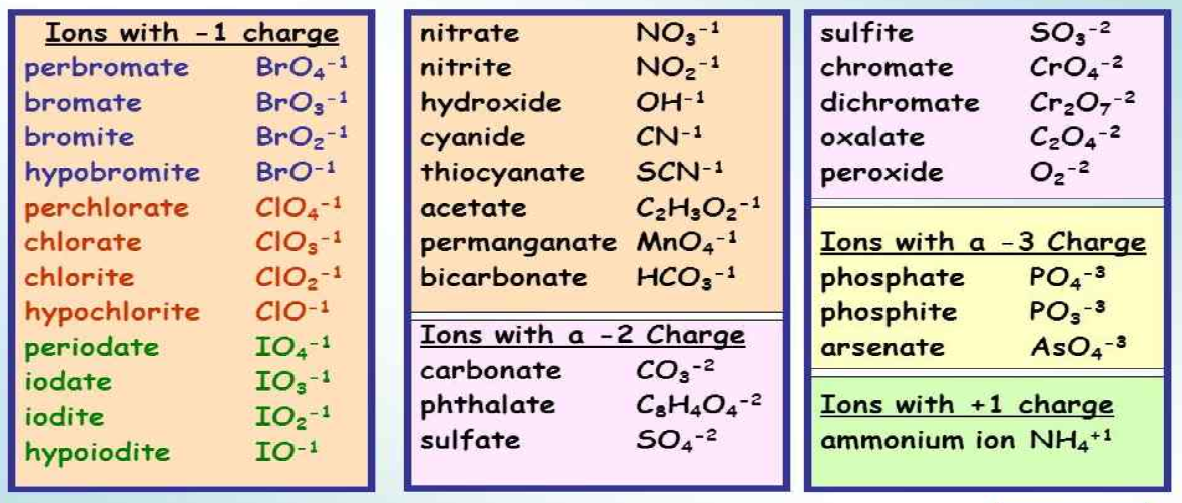
Ion Name Common Polyatomic Ions (Alphabetical order by ion name) NOTE: -ite ending means one less oxygen than the -ate form. Ion Name Acetate Ammonium Arsenate Arsenite Azide Borate Bromate Bromite Carbonate Chlorate Chlorite Ion Symbol CH3CO2- or CH3COO- NH4 + AsO4 3- AsO33- N3 - BO3 2- BrO3 - BrO2 - CO3 2- ClO3- ClO2- Ion Name Dichromate Google Classroom Learn what polyatomic ions are and how they bond. Some ions consist of a single atom with a net charge. They're called monatomic ions. Examples include Na + , O 2 − , and Cl − . Other ions consist of a molecule —a group of atoms covalently bonded together—with a net charge. They're called polyatomic ions. Common Polyatomic Ions Name(s) Formula Name(s) Formula ammonium NH4 + acetate CH3COO-C2H3O2-bromate BrO3-carbonate CO3 2-chlorate ClO3-chlorite ClO2-chromate CrO4 2-cyanide CN-dichromate Cr2O7 2-hydrogen carbonate bicarbonate HCO3-hydrogen sulfate bisulfate HSO4-hydrogen phosphate biphosphate HPO4 2-hydroxide OH- A polyatomic ion (also known as a molecular ion) is a covalent bonded set of two or more atoms, or of a metal complex, that can be considered to behave as a single unit and that has a net charge that is not zero. [1] The term molecule may or may not be used to refer to a polyatomic ion, depending on the definition used.
Polyatomic Ions Naming and Formulas Study Guide Inspirit
Polyatomic ions are formed when a group of atoms have a charge. Hydroxide, for example, is formed when oxygen and hydrogen covalently bond but still have a charge of -1. When a polyatomic ion forms an ionic bond with another ion, a polyatomic ionic compound is made. For example, the +1 barium ion can form an ionic bond with the -1 hydroxide ion, to form the Barium Hydroxide (BaOH) ionic compound. Table of contents List of polyatomic ions List of monatomic ions FAQs List of polyatomic ions Ions made up of more than one atom are known as polyatomic ions. Ions with positive charge are called cations. Ions with negative charge are called anions. List of monatomic ions The ions made of a single atom are called simple ions or monatomic ions.
Table 3.4.1 3.4. 1 lists the ion names and ion formulas of the most common polyatomic ions. For example, NO−3 NO 3 − is the nitrate ion; it has one nitrogen atom and three oxygen atoms and an overall 1− charge. Note that there are two polyatomic ions in this table, the ammonium ion and hydronium ion that are cations. The ions that we have discussed so far are called monatomic ions, that is, they are ions formed from only one atom. We also find many polyatomic ions. These ions, which act as discrete units, are electrically charged molecules (a group of bonded atoms with an overall charge). Some of the more important polyatomic ions are listed in Table 2.5.
PolyatomicIons Pathways to Chemistry
♦ Polyatomic Ions - A group of atoms held together by covalent bonds found in ionic compounds. ♦ Know/ memorize/ recognize names, formulas and charges! General Information: Recognizing Ionic vs. Covalent Compounds: Compounds Compounds Polyatomic ions are ions that contain more than one element. This polyatomic ions list contains many common ions, grouped by charge. Each entry contains the ion's name, molecular formula and chemical structure. +1 Polyatomic Ions -1 Polyatomic Ions -2 Polyatomic Ions -3 Polyatomic Ions




