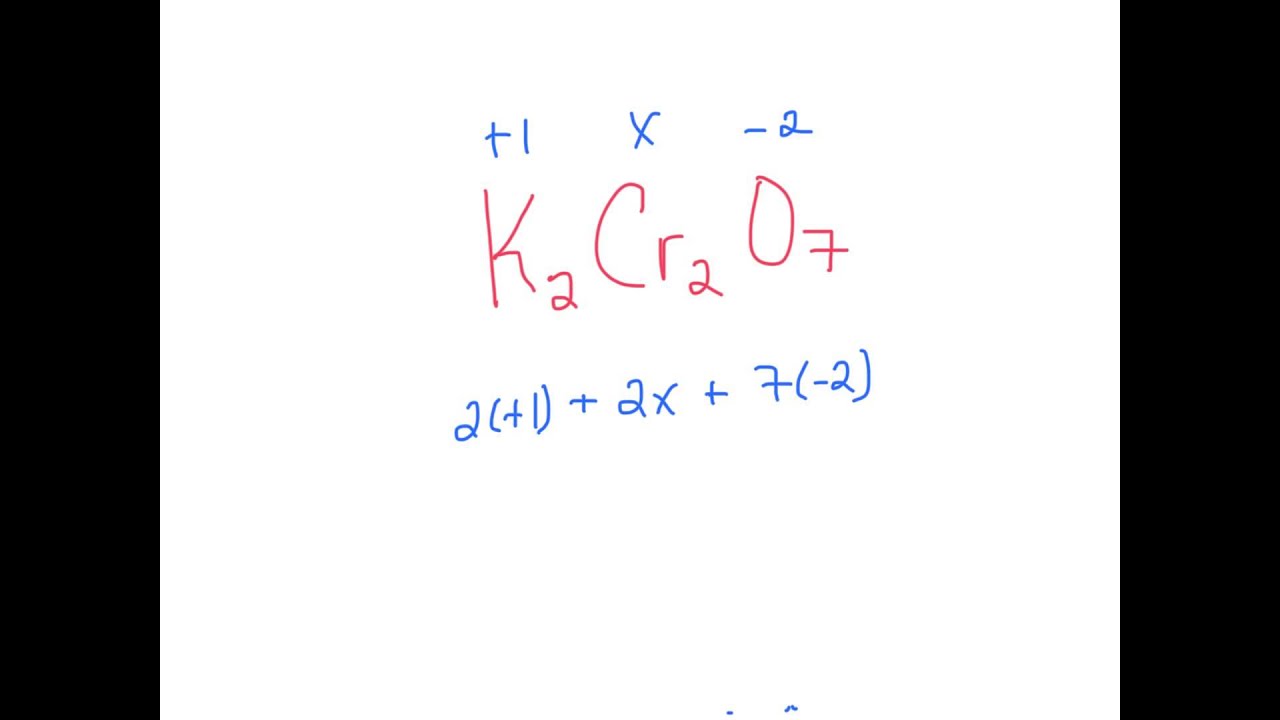Step 4: Substitute Coefficients and Verify Result. Count the number of atoms of each element on each side of the equation and verify that all elements and electrons (if there are charges/ions) are balanced. 4 K2Cr2O7 + 3 H2S + 13 H2SO4 = 4 K2SO4 + 4 Cr2(SO4)3 + 16 H2O. Reactants. About K2Cr2O7 H2S H2SO4. Potassium dichromate (K2Cr2O7) is a common chemical inorganic reagent most commonly used as an oxidizing agent in various laboratories. It's a crystalline ionic strong, very bright, red-orange. Among chemists, Potassium Dichromate is very common in determining the unknown concentration of secondary standard substances solution.

Napisz reakcje redoks, wskaż reduktor, utleniacz i zapisz reakcje połówkowe K2Cr2O7 + H2S
Solved and balanced chemical equation K2Cr2O7 + 3 H2S + 4 H2SO4 → K2SO4 + Cr2(SO4)3 + 3 S + 7 H2O with completed products. Application for completing products and balancing equations.. [Note: Sense of smell becomes rapidly fatigued & can NOT be relied upon to warn of the continuous presence of H2S. Shipped as a liquefied compressed gas.] $$\ce{H2S + K2Cr2O7 + 4H2SO4 -> SO2 + Cr2(SO4)3 + K2SO4 + 5H2O}?\label{rxn:QR2}\tag{R2}$$ The reaction seems stoichiometrically plausible, yet likely requires highly concentrated oxidants. I know \eqref{rxn:QR1} happens at low concentration of $\ce{H2SO4}$. Maybe using highly concentrated $\ce{H2SO4}$ would do the work for \eqref{rxn:QR2}. What. Detailed information about the equation. Reaction conditions when applied H2S + H2SO4 + K2Cr2O7. Reaction process H2S + H2SO4 + K2Cr2O7. The result of the reaction H2S + H2SO4 + K2Cr2O7. Balance Chemical Equation - Online Balancer. Word equation; 4 Potassium dichromate + 3 Sulfane + 13 Sulfuric acid = 4 Potassium sulfate + 4 Chromium(III) sulfate + 16 Water

Презентация по Химии "Хром" скачать смотреть бесплатно
This video is the practical demonstration of the reaction of Acidified Potassium dichromate (k2Cr2O7+H2SO4) with Hydrogen peroxide (H2O2).Precipitation and d. Step 4: Substitute Coefficients and Verify Result. Count the number of atoms of each element on each side of the equation and verify that all elements and electrons (if there are charges/ions) are balanced. K2Cr2O7 + 4 H2So4 + 3 H2S = K2So4 + Cr2(So4)3 + 7 H2O + 3 S. Reactants. Here's what I got. Start by writing the unbalanced chemical equation. Potassium dichromate, "K"_2"Cr"_2"O"_7, will react with hydrosulfuric acid, which is aqueous. Step 4: Substitute Coefficients and Verify Result. Count the number of atoms of each element on each side of the equation and verify that all elements and electrons (if there are charges/ions) are balanced. 4 K2Cr2O7 + -3 H2S + 15 H2SO4 = 4 Cr2(SO4)3 + -8 S + 8 KHSO4 + 8 H2O. Reactants. Products.
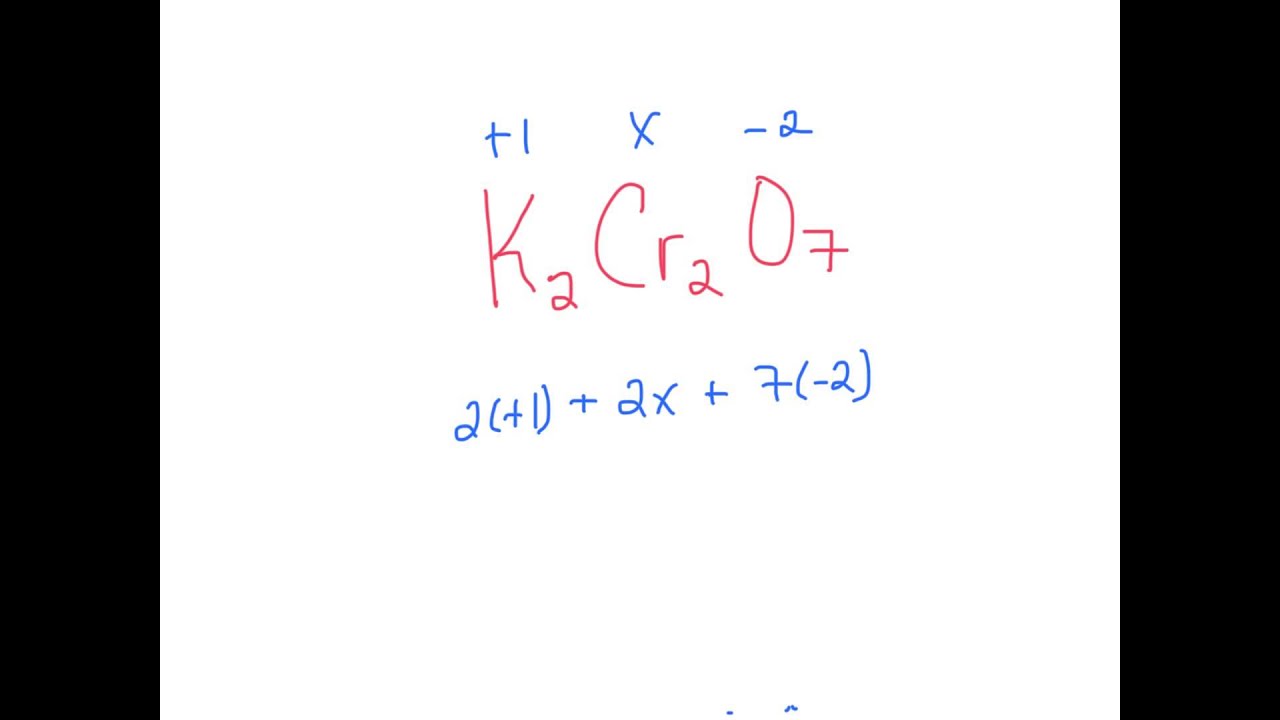
K2Cr2O7 YouTube
K2Cr2O7+H2SO4+SO2=K2SO4+Cr2(SO4)3+H2O balance the redox reaction by ion electron method or half reaction method. k2cr2o7+h2so4+so2=k2so4+cr2(so4)3+h2o. This reaction can be represented by the following equation: K2Cr2O7 + H2SO4 → H2CrO4 + K2SO4. Chromic acid is a strong oxidizing agent and plays a crucial role in the subsequent steps of the reaction. Oxidation of Sulfuric Acid: In the presence of chromic acid, sulfuric acid gets oxidized to form sulfur trioxide (SO3).
Balance the chemical equation k2cr2o7+h2so4+so2=k2so4+cr2(so4)3+H2O Balance the chemical reaction K2Cr2O7+H2SO4+SO2=K2SO4+Cr2(SO4)3+H2O Potassium dicro. Step 1. Write the skeleton equation. The molecular equation is. K2Cr2O7 + H2SO4 +SO2 → K2SO4 + Cr2(SO4)3 +H2O. Strip the equation of all common ions, also H+,OH- and H2O (these come back in during the balancing procedure). K+ + Cr2O2- 7 + H+ + SO2- 4 + SO2 → K+ + SO2- 4 +Cr3+ + SO2- 4 + H2O. The above equation simplifies to. Cr2O2- 7 +SO2.

1.Oblicz stopnie utlenienia pierwiastków w następujących związkach SiO2, MgO, H2S, NH3, H3PO4
Step 4: Substitute Coefficients and Verify Result. Count the number of atoms of each element on each side of the equation and verify that all elements and electrons (if there are charges/ions) are balanced. 0 K2Cr2O7 + 3 H2S + H2SO4 = 0 Cr2(SO4)3 + 0 K2SO4 + 4 S + 4 H2O. Reactants. Products. Solved and balanced chemical equation K2Cr2O7 + 3 H2O2 + 4 H2SO4 → Cr2(SO4)3 + 7 H2O + K2SO4 + 3 O2 with completed products. Application for completing products and balancing equations. Chemical Equations online! Submit. Advanced search. K 2 Cr 2 O 7 + 3 H 2 O 2 + 4 H 2 S O 4 → Cr 2 (S O 4) 3 + 7 H 2 O + K 2 S O 4 + 3 O 2. This.