Chemical Formula Definition: An expression which states the number and type of atoms present in a molecule of a substance. Chemical formulas such as HClO4 can be divided into empirical formula, molecular formula, and structural formula. Types of Chemical Reactions Dissolution Precipitation Acids and Bases and their reactions Oxidation-Reduction Reactions Dissolution of Ionic Compounds Dissolution of Molecules Electrolytes Substances which increase the conductivity of water when they dissolve

HelpWork balancing chemistry equations
Microsoft Word - Chapter 6.doc. 82. Returning to our earlier, mistake free equation: CH4 + O2. →. CO2 + H2O. We are ready for the second absolute requirement: "The number of atoms of each. element must be the same on both sides of the equation.". In our equation, we have. A chemical equation describes what happens in a chemical reaction. The equation identifies the reactants (starting materials) and products (resulting substance), the formulas of the participants, the phases of the participants (solid, liquid, gas), and the amount of each substance. CHAPTER1 C onsider the following situations of daily life and think what happens when - milk is left at room temperature during summers. an iron tawa/pan/nail is left exposed to humid atmosphere. grapes get fermented. food is cooked. food gets digested in our body. we respire. A chemical reaction is described by a chemical equation, an expression that gives the identities and quantities of the substances involved in a reaction. A chemical equation shows the starting compound (s)—the reactants —on the left and the final compound (s)—the products —on the right, separated by an arrow.
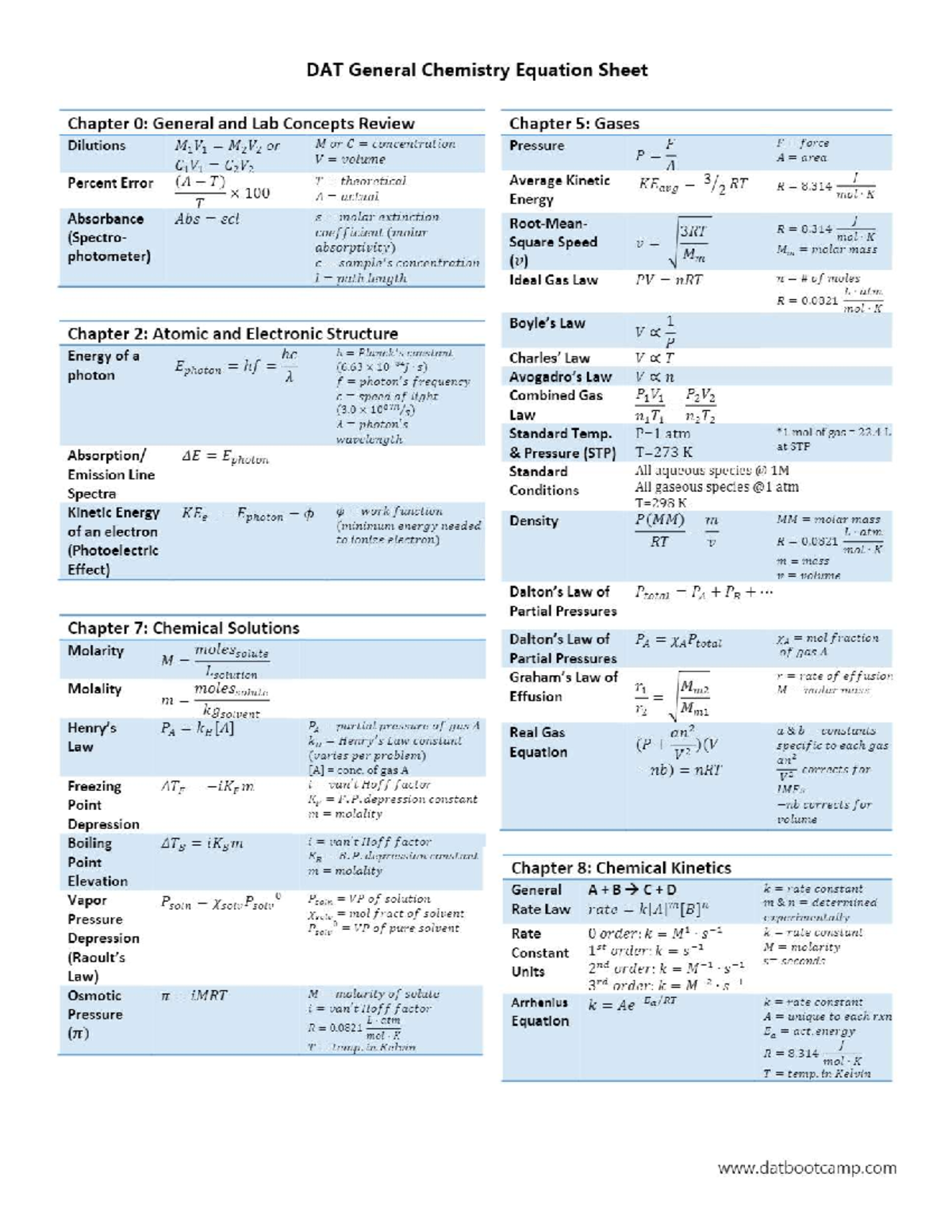
Chem equation sheet for chemistry class Eng Tech 1Ch3 McMaster StuDocu
Chapter 11. Chemical Equations 11.1. ChemicalEquation Concepts Under the right circumstances substances change into newsubstances. Suchchanges are calledchemical transformations,orreactions.The original substances are calledreac- tants,the substances resulting from the change are calledproducts.Chemical reactions reveal the basic properties of substances that form the heart of chemistry. The chemical equation described in section 4.1 is balanced, meaning that equal numbers of atoms for each element involved in the reaction are represented on the reactant and product sides. This is a requirement the equation must satisfy to be consistent with the law of conservation of matter. chemical reactions when the names of the reactants and products are specified; 1.5.8 write balanced ionic equations for reactions covered in this specification; 1.5.9 write half equations for reactions covered in this specification; 1.5.10 demonstrate knowledge and understanding that in chemical equations the three states of matter Chemical equations and formulas are a vital part of learning chemistry from 11‒19. Being able to use them correctly is part of what it means to be a chemist. Formulas and equations allow chemists to communicate chemical knowledge efficiently. Students should understand that chemical formulas:
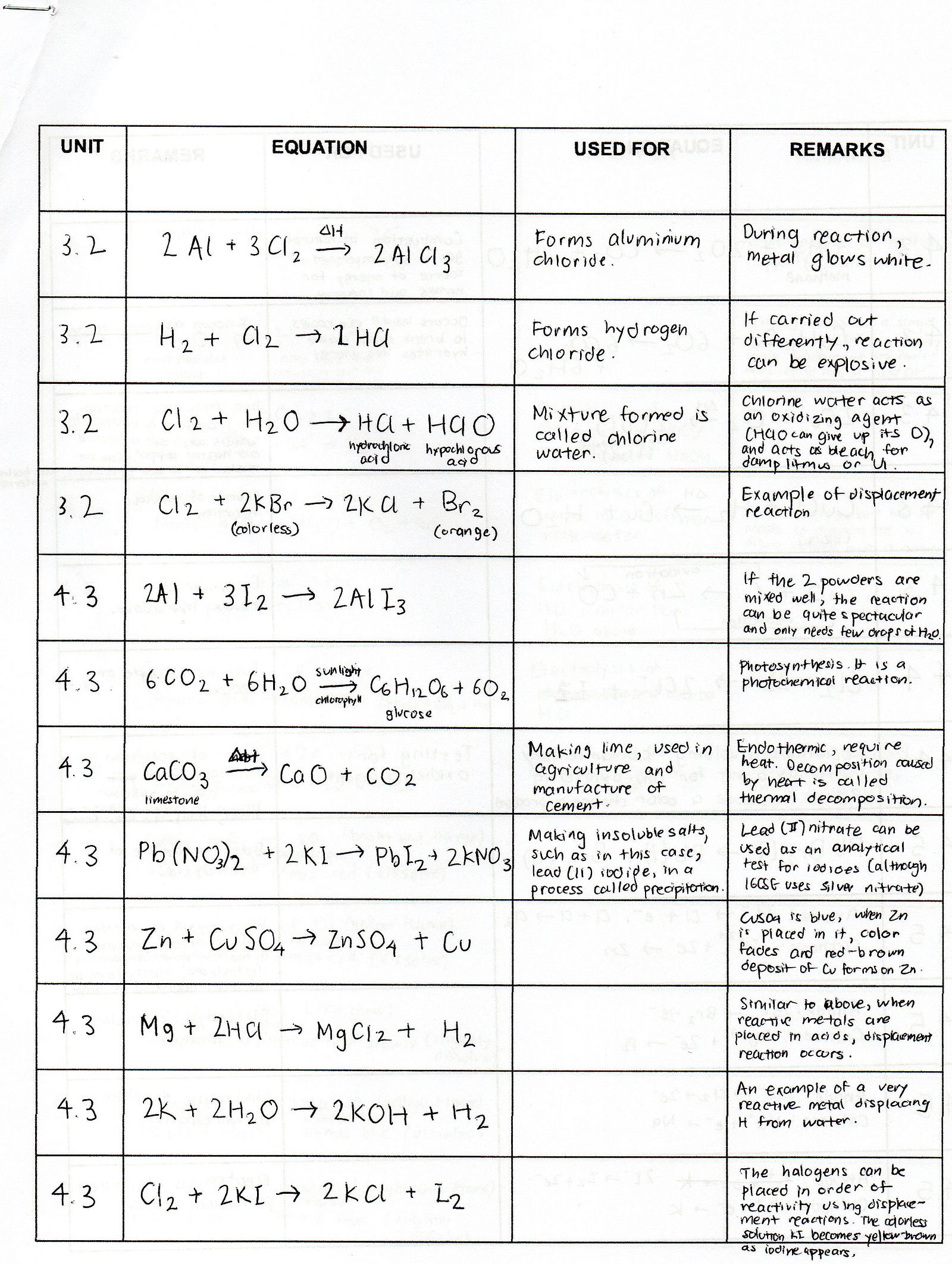
Chemistry Formulas And Equations Review Tessshebaylo
1 atom Fe 2 atoms O Right side: 2 atoms Fe 3 atoms O The balancing of the equation is accomplished by introducing the proper number or coefficient before each formula. To balance the number of O atoms, write a 3 in from of the O2 and a 2 in front of the Fe2O3: Fe + 3 O2 → 2 Fe2O3 4.4 Chemical changes 43 4.5 Energy changes 51 4.6 The rate and extent of chemical change 55 4.7 Organic chemistry 61 4.8 Chemical analysis 70 4.9 Chemistry of the atmosphere 75 4.10 Using resources 80 4.11 Key ideas 89 5 Scheme of assessment 91 5.1 Aims and learning outcomes 91 5.2 Assessment objectives 92 5.3 Assessment weightings 93
The general equation for a chemical reaction is: As to why water is produced as a gas, it may be observed that the water is produced as a result of an explosion as shown in Figure 7.3.1 7.3. 1. At the elevated temperature of the explosion, water exists as a gas and not as a liquid. OBJECTIVES After completing this lesson, you will be able to: write and balance simple chemical equations; describe the significance of a balanced chemical equation; explore the relationship between mole, mass and volume of various reactants and products;

Chemical Equations PDF
CHAPTER 1 WHAT ARE CHEMICAL ENGINEERING AND BIOENGINEERING? 3 1.1 Introduction 3 1.2 A Brief History of Chemical Engineering 4. 7.2 Real Gases: Equations of State 366 7.3 Real Gases: Compressibility Charts 377 7.4 Real Gas Mixtures 384 CHAPTER 8 MULTIPHASE EQUILIBRIUM 411 To indicate more than one polyatomic ion in a chemical formula, place parentheses around the polyatomic ion and use a subscript. Roman numerals indicate the oxidation number of cations having multiple possible oxidation states. After reading Lesson 9.2, answer the following questions. Binary Ionic Compounds 1.



