329 Share 110K views 10 years ago A quick explanation of the molecular geometry of CO3 2- including a description of the CO3 2- bond angles..more.more A quick explanation of the. Step 1: Count the Total Number of Valence Electrons. In CO32- ion, we have one carbon atom and three oxygen atoms along with two negatively charged electrons carrying the charge. Valence electrons refer to the number of electrons in the outermost shell of an atom around the nucleus that help in determining the valency of the given atom.
Co Formulacion SEO POSITIVO
1.8K 161K views 3 years ago This chemistry video tutorial explains how to draw the lewis structure of CO3 2- also known as the carbonate ion. This video discusses the resonance structure of. A step-by-step explanation of how to draw the CO3 2- Lewis Dot Structure (Carbonate ion). For the CO3 2- structure use the periodic table to find the total number of valence electrons. Figure 5.2.9 5.2. 9: (a) H2O has four regions of electron density around the central atom, so it has a tetrahedral electron-pair geometry. (b) Two of the electron regions are lone pairs, so the molecular structure is bent. Exercise 5.2.3 5.2. 3. The hydronium ion, H 3 O +, forms when acids are dissolved in water. CO3 2- Lewis Structure Step-by-Step Guide 1. Determine the total number of valence electrons In the carbonate ion (CO3^2-), carbon (C) contributes 4 valence electrons, while each oxygen (O) atom contributes 6 valence electrons. Since there are three oxygen atoms, the total number of valence electrons is:
¿Cuál es la estructura de Lewis de Co3 2?
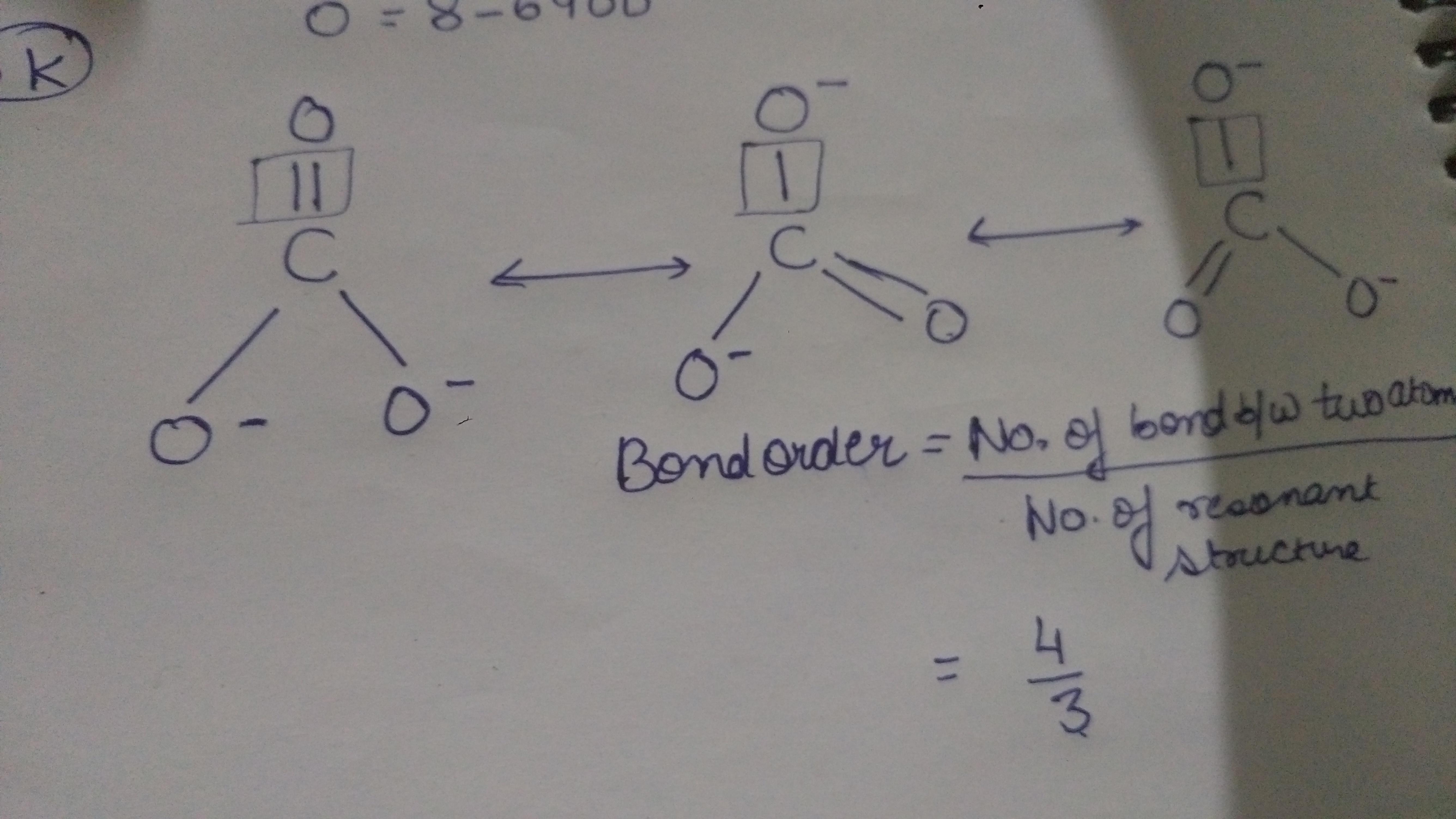
For the CO 32- Lewis structure there are a total of 24 valence electrons available. Transcript: Let's do the CO3 2- Lewis structure: the carbonate ion. Carbon has 4 valence electrons; Oxygen has six, we have 3 Oxygens, and this negative 2 means we have an extra two valence electrons. Add that all up: 4 plus 18 plus 2: 24 valence electrons. Lewis structure of CO3 2- contains one double bond and two single bonds between the Carbon (C) atom and Oxygen (O) atom. The Carbon atom (C) is at the center and it is surrounded by 3 Oxygen atoms (O). Both the single bonded Oxygen atoms (O) have -1 formal charge. Let's draw and understand this lewis dot structure step by step. Lewis structure of carbonate ion is drawn in this tutorial step by step. Total valence electrons concept is used to draw the lewis structure of CO 32-. After finishing the lewis structure of CO 32-, there should be a -2 charge and it should be stabile structure. You will learn about these facts in this tutorial. Carbonate ion | CO 32- CO32- Geometry and Hybridization - Chemistry Steps Examples Geometry CO32- Geometry and Hybridization There are 4 + 3×6 + 2 = 24 electrons. The carbon goes in the middle, and the oxygens take 6 electrons each as three lone pairs: The carbon lacks an octet, so we use a lone pair from one oxygen to make a double with it.
What are some examples of trigonal sp^2 hybrids? Socratic
I quickly take you through how to draw the Lewis Structure of CO3 2- (Carbonate Ion). I also go over the resonance, hybridization, shape and bond angle. The first step in drawing the CO 3 2-Lewis structure is to determine the total number of valence electrons in the molecule. This can be calculated by multiplying the valence electrons of each atom. Carbon is located in group 14 of the periodic table and has four valence electrons, while oxygen, belonging to group 16, has six valence electrons. In CO 3 2-, which consists of one carbon atom and.
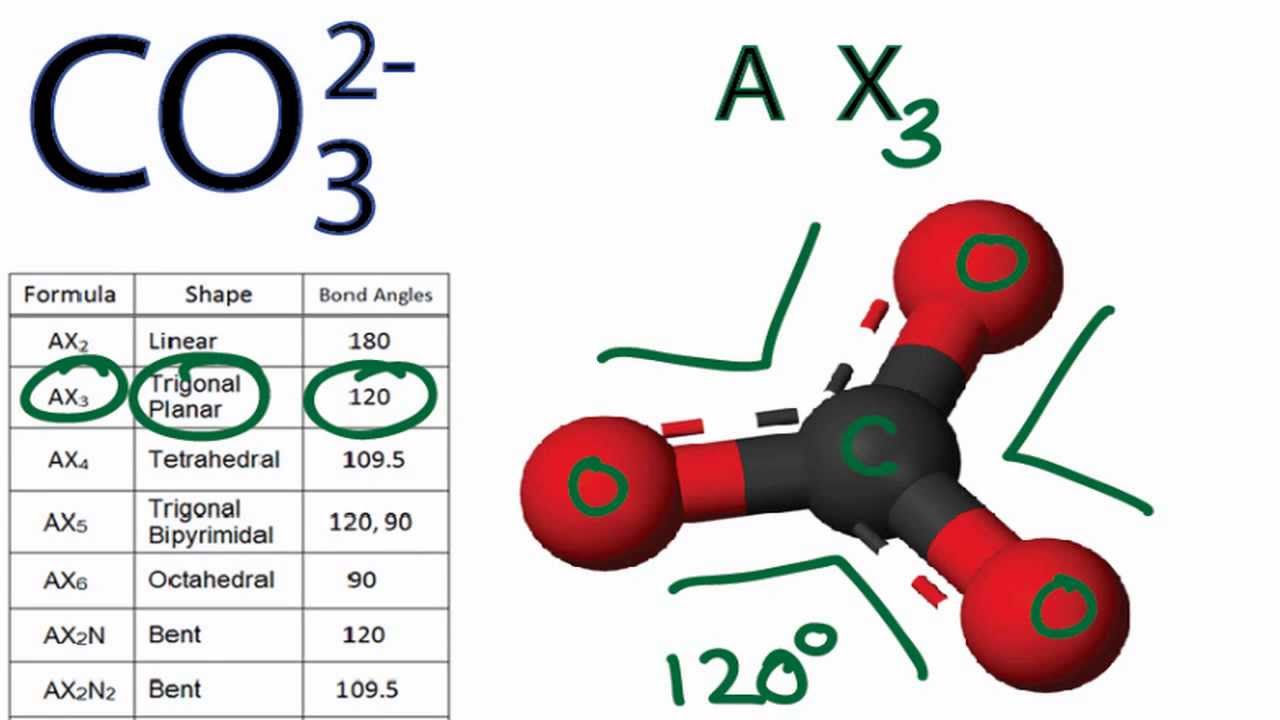
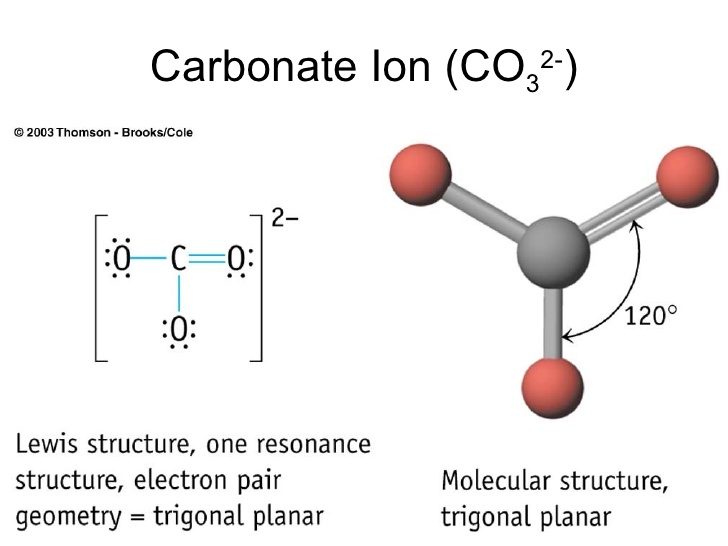
Carbonate, \(\ce{CO3^2-}\), is a common polyatomic ion found in various materials from eggshells to antacids. What are the electron-pair geometry and molecular structure of this polyatomic ion? Answer. The electron-pair geometry is trigonal planar and the molecular structure is trigonal planar. Due to resonance, all three C-O bonds are identical. 1. Count the total valence electrons in [CO3]2- The Lewis dot structure of a molecule is referred to as a simplified representation of all the valence electrons present in it. Therefore, the very first step while drawing the Lewis structure of [CO 3] 2- is to count the total valence electrons present in the concerned elemental atoms.

[Download 35+] Possible Resonance Structures For Co32
The Lewis structure of H 2 O indicates that there are four regions of high electron density around the oxygen atom: two lone pairs and two chemical bonds: Figure 4.3.9 4.3. 9. Thus, the electron-pair geometry is tetrahedral and the molecular structure is bent with an angle slightly less than 109.5°. Step - 8 Last is to determine shape, hybridization and bond angle of CO32- lewis structure. CO32- lewis structure.. Carbonate (CO32-) ions have 2- negative formal charge and also it has quite sufficient lone electron pairs present on three O atoms out if which two O atoms have -1 negative charge. Thus it can easily gain or accepts H+ ions.




