ICl3, named Iodine Trichloride, is an Interhalogen compound. Interhalogen compounds are molecules, which contain at least two different halogen atoms. These compounds are generally written as ABn where n= 1, 3, 5, and 7; A and B are less electronegative and more electronegative elements, respectively. An explanation of the molecular geometry for the ICl3 (Iodine trichloride) including a description of the ICl3 bond angles. The electron geometry for the Iod.

The Best 29 Icl3 Polar Or Nonpolar imageyardask
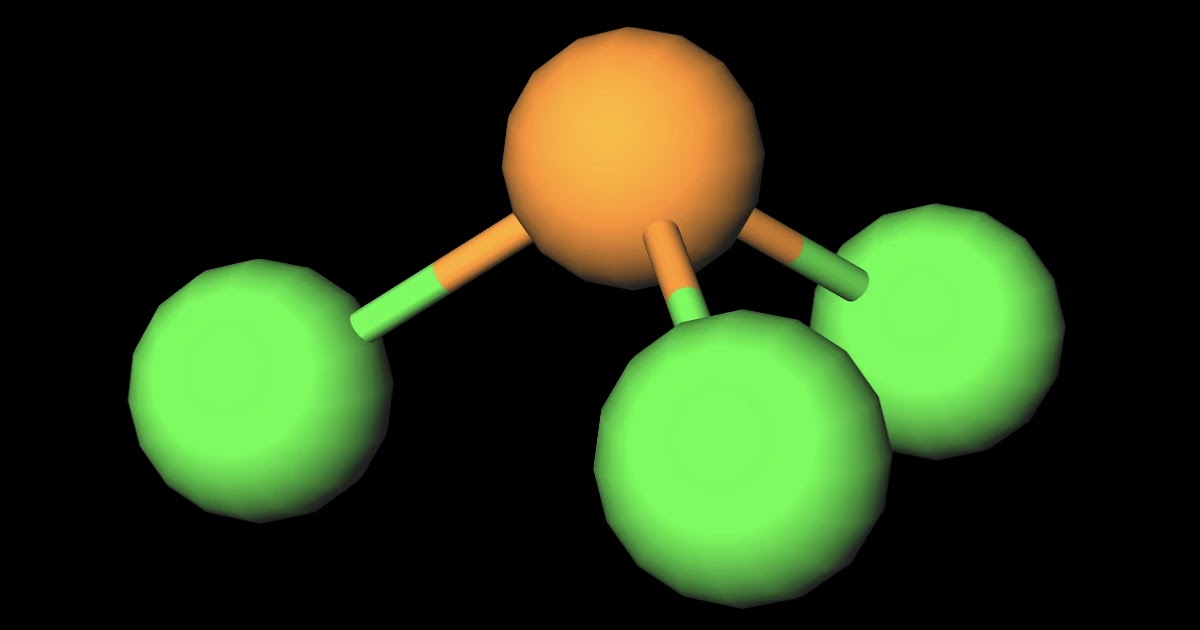
ICl3 is a polar molecule because it has poles of partial positive charge (ẟ+) and partial negative charge (ẟ-) on it. Let me explain this to you in 3 steps! Step #1: Draw the lewis structure Here is a skeleton of ICl3 lewis structure and it contains three I-Cl bonds. Which of these molecules and ions contain polar bonds? Which of these molecules and ions have dipole moments?ICl3OpenStax™ is a registered trademark, which w. Iodine trichloride (ICl3) is a polar molecule. The central iodine (I) atom in the ICl3 molecule is surrounded by three chlorine (Cl) atoms via single covalent bonds, forming an asymmetric T-shaped molecule. The electronegativity of the chlorine (Cl) atom is greater than the iodine (I) atom. Chemistry Chemistry questions and answers Draw the lewis structure for ICl3 (iodine trichloride). What is the electron geometry and molecular geometry (shape) of the molecule? Refer to the provided chart to aid you in your answer. Is the molecule polar or non polar? This problem has been solved!

The Best 29 Icl3 Polar Or Nonpolar imageyardask
ICl3) molecule is classified as a polar molecule. The molecule of iodine trichloride (with trigonal bipyramidal molecular geometry) is tilted, the bond angles between iodine and. Molecules can be classified as polar or nonpolar. The molecule polar behaves in a different manner as compared to nonpolar. Overview: Drawing the Lewis Structure for ICl 3. In the ICl 3 Lewis structure Iodine (I) is the least electronegative atom and goes in the center of the Lewis structure. The ICl 3 Lewis structure you'll need to put more than eight valence electrons on the Iodine atom. In the Lewis structure for ICl 3 there are a total of 28 valence electrons. ICl3 molecule is made of one iodine, three chlorine atoms. The chlorine and iodine atoms have s and p orbitals. Chlorine comes as the second element from the halogen family in the periodic table. The iodine atom also belongs to the same family group. But it falls as the fourth element in the periodic table. The molecule is symmetric. The two oxygen atoms pull on the electrons by exactly the same amount. Propane is nonpolar, because it is symmetric, with H atoms bonded to every side around the central atoms and no unshared pairs of electrons. Exercise 4.12. 1. Label each of the following as polar or nonpolar.

Is ICL3 polar? chemistry
A small difference (<0.4) results in a nonpolar covalent bond, an intermediate difference (0.4 - 1.7) results in a polar covalent bond, and a large difference (>1.7) results in an ionic bond. According to the figure above, a difference in electronegativity (\(\Delta\) EN) greater than 1.7 results in a bond that is mostly ionic in character. Explain how a molecule that contains polar bonds can be nonpolar. Answer. As long as the polar bonds are compensated (for example. two identical atoms are found directly across the central atom from one another), the molecule can be nonpolar. PROBLEM \(\PageIndex{2}\)
A. Is ICl3 polar or nonpolar? B. Is PF polar or nonpolar? C. Is SF6 polar or nonpolar? Submit Answer Try Another Version 1 item attempt remaining pl PE ICl3 has a trigonal bipyramid structure and it is highly symmetrical. The Cl are in the in the x,y plane in a radial symmetry and the both lone pairs are in z plane in opposition of each other. So all of the dipole moments created by the bonds have a net zero direction because of symmetric opposition. 6
MakeTheBrainHappy Is PCl3 Polar or Nonpolar?
ICl or iodine monochloride is an interhalogen compound which is formed by reaction with chlorine and iodine. It's boiling point and melting point is 97.40C and 27.20C respectively. It is a reddish brown colored substance which is corrosive in nature. Iodine chloride is used as source of electrophilic iodine, preparing iodates. The chemical name of the $ IC {l_3} $ molecule is Iodine trichloride. It is an interhalogen compound made up of iodine and chlorine. Iodine has seven electrons and each chlorine provides one electron giving a total of five pairs.




