Apply a stoichiometric conversion factor to convert between the molar quantities of two substances that participate in a chemical reaction.. Recall that, when using a calculator, each conversion factor should be entered in parentheses, or the "=" key should be used after each division. In this case, 7.53 × (1 mol O 2 ÷ 2). This stoichiometry calculator lets you calculate the relative amounts of reactants and products involved in a chemical reaction.
J² + H = Element 119 Kariodisonium Stoichiometry Calculations
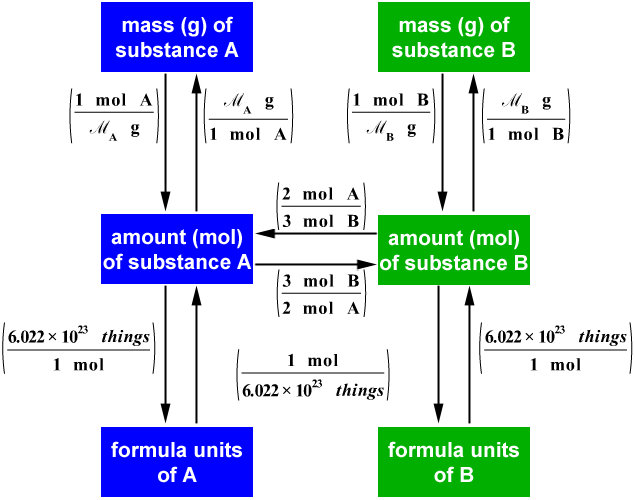
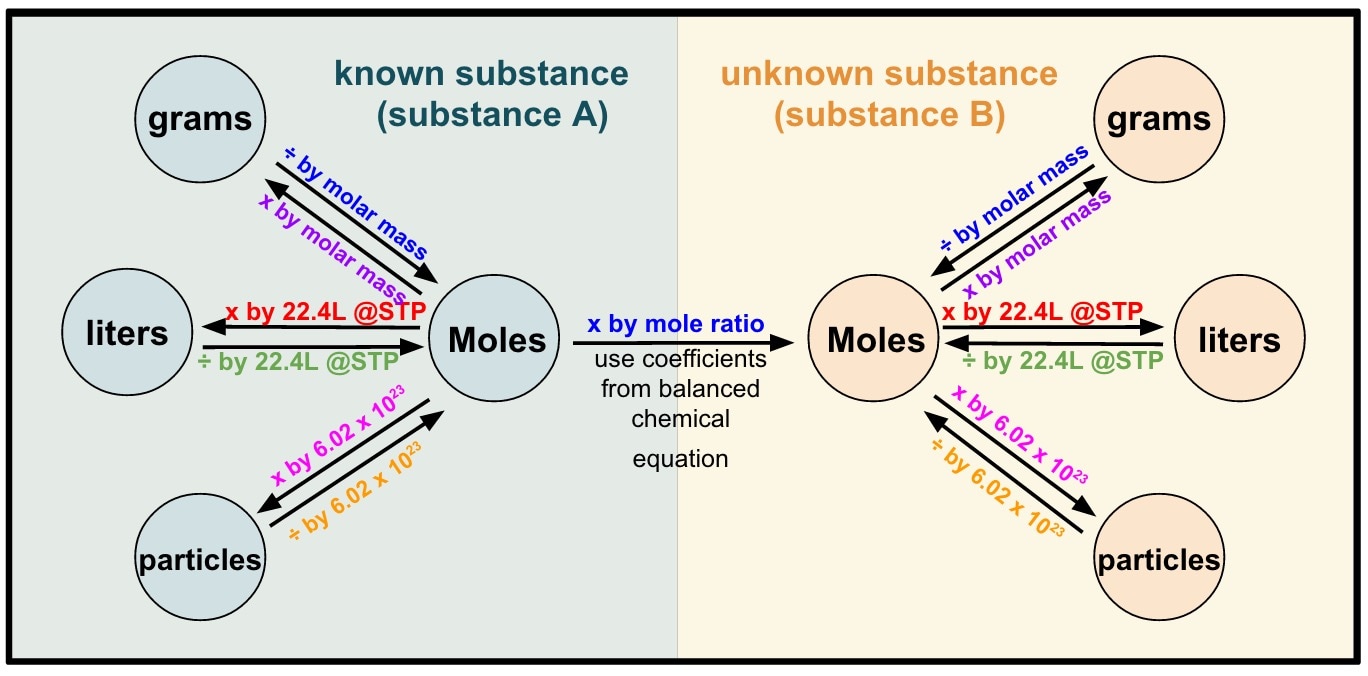
Figure \(\PageIndex{1}\): A Flowchart for Stoichiometric Calculations Involving Pure Substances. The molar masses of the reactants and the products are used as conversion factors so that you can calculate the mass of product from the mass of reactant and vice versa. Flowchart of steps in stoichiometric calculations. To perform a stoichiometric calculation, enter an equation of a chemical reaction and press the Start button. The reactants and products, along with their coefficients will appear above. Enter any known value. The remaining values will automatically be calculated. Conversion factors from a chemical equation. Stoichiometry pronounced as "stɔɪkiˈɒmɪtri" is the calculation of the amount of reactants and products in a chemical reaction. It is based on the fact that a balanced chemical equation is also a set of mole-to-mole equalities between the reactants and the products. Each equality gives two. 1 mol Fe 2 O 3: 2 mol Al Using this ratio, we could calculate how many moles of Al are needed to fully react with a certain amount of Fe 2 O 3 , or vice versa. In general, mole ratios can be used to convert between amounts of any two substances involved in a chemical reaction.
Stoichiometry Review Mr. Siemianowski Eisenhower High School
Stoichiometry is the field of chemistry that is concerned with the relative quantities of reactants and products in chemical reactions. For any balanced chemical reaction, whole numbers (coefficients) are used to show the quantities (generally in moles ) of both the reactants and products. For example, when oxygen and hydrogen react to produce. Stoichiometry Tutorials: Dimensional Analysis / Stoichiometric Conversions (from a complete OLI stoichiometry course) Dimensional analysis allows us to change the units used to express a value. For instance, it allows us to convert between volume expressed in liters and volume expressed in gallons. Using Stoichiometry in Conversions September 13th, 2011 | Author: Andrew We can further expand our understanding of stoichiometry by using the system of ratios we have created as conversions for questions that we are asked in chemistry. Stoichiometry. We shall set up Stoichiometric Tables using A as our basis of calculation in the following reaction. We will use the stoichiometric tables to express the concentration as a function of conversion. We will combine C i = f(X) with the appropriate rate law to obtain -r A = f(X). Topics. Batch System Stoichiometric Table

Stoichiometry Chemistry Activities
Chapter 3: Stoichiometry Conversions Conversions Procedure Step 1: Determine where you are starting and finishing Step 2: Start calculation by writing down what you know Step 3: Use dimensional analysis Set up problem so you can cancel out units Tools Available Avogadro's Constant 6.022×10 6 7 - 𝑜 Periodic Table (M) Stoichiometry is simply the math behind chemistry. Given enough information, one can use stoichiometry to calculate masses, moles, and percents within a chemical equation.. Once we know the moles of solute we can look at the periodic table and figure out the conversion from moles to grams. Molality = moles solute kg solvent: Now we simply.
3Ag(s) + Al(NO 3) 3(aq) → 3AgNO 3 + Al(s) Solution. Here, we first must convert the mass of Ag to moles before using the balanced chemical equation and then the definition of molarity as a conversion factor: 3.66 gAg × 1 molAg 107.97 gAg × 1 molAl(NO3)3 3 molAg ×1Lsolution0.0995. The strikeouts show how the units cancel. A comprehensive reaction stoichiometry calculator that can solve problems of all situations. It automatically balances equations and finds limiting reagents. It can also handle equations that contains fractions and decimals.
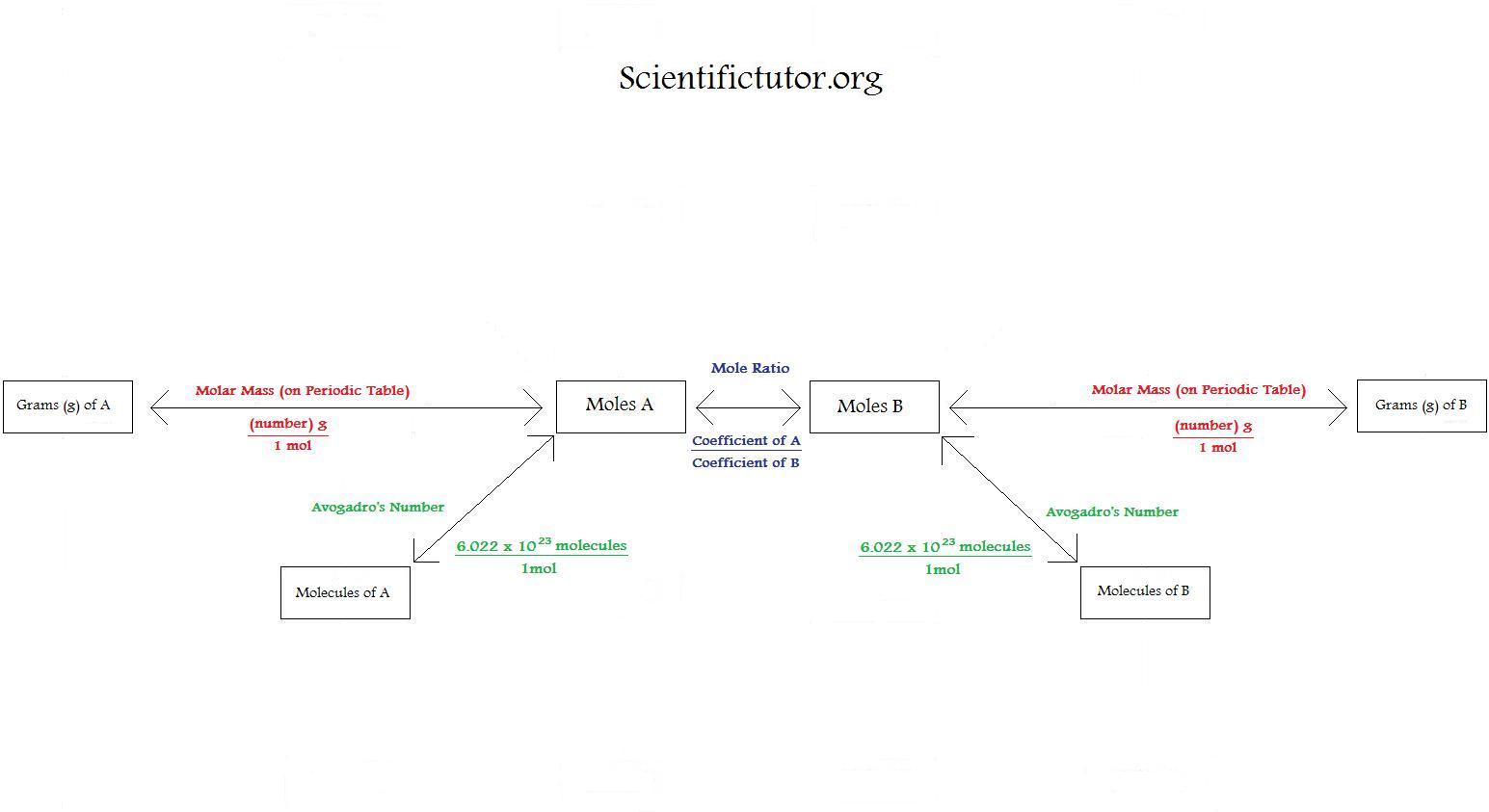
Using Stoichiometry in Conversions Scientific Tutor
Multiply wt% ELEMENT by numerical value below for equivalent expressed as OXIDE. Element. Oxide. Factor. Silver. Ag 2 O. 1.0741. Aluminium. This online Stoichiometry Calculator finds the stoichiometric coefficients to balance a given chemical equation and computes amounts of the reactants and products of the reaction, both in moles and grams. The equations may include free electrons and electrically charged molecules (ions) as well as hydrated compounds.




