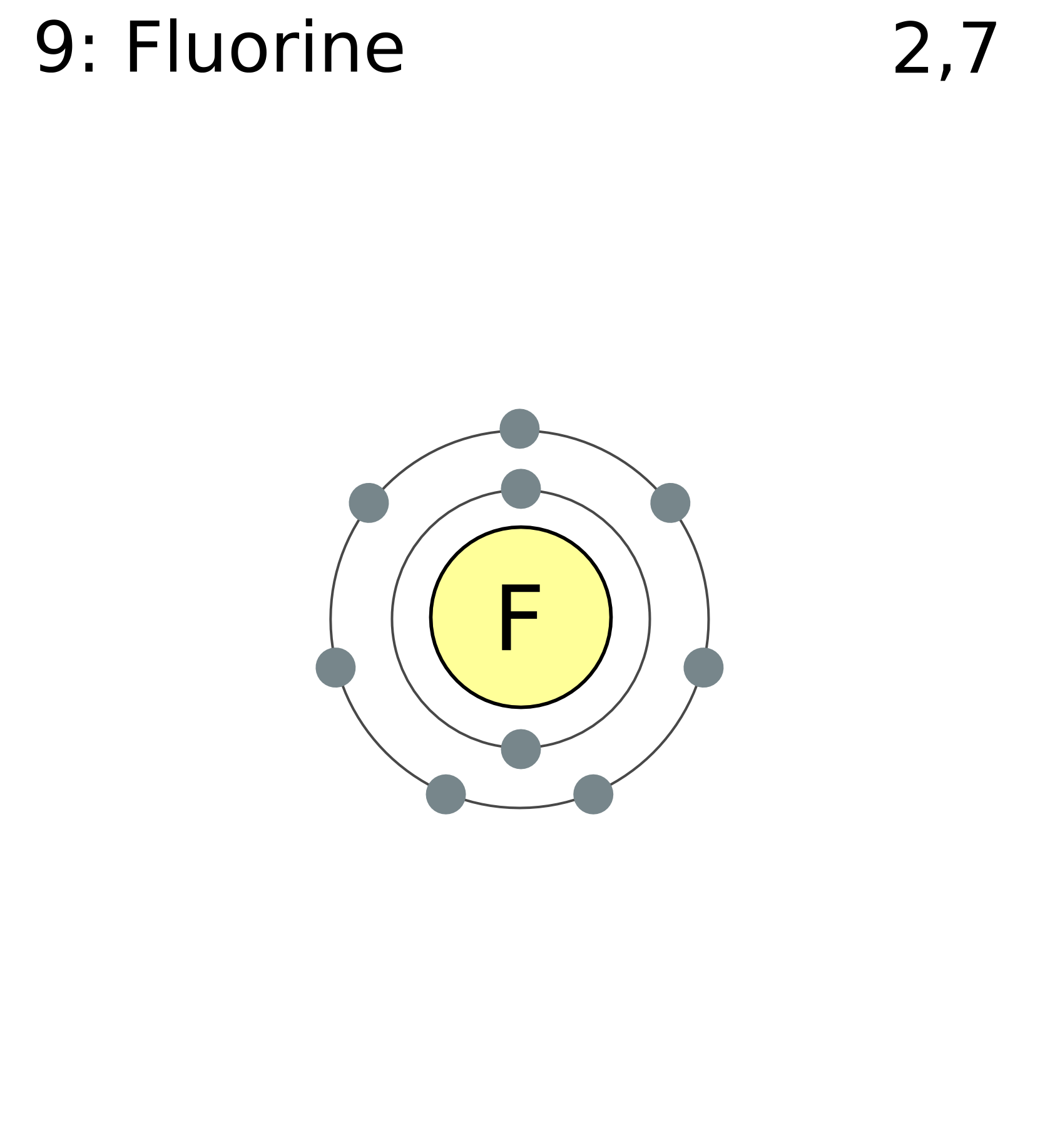
Fluorine has 2 electrons in its first shell and 7 in its second.Check me out: http://www.chemistnate.com In this video we'll look at the atomic structure and Bohr model for the Fluorine atom (F). We'll use a Bohr diagram to visually represent where the electrons.
Fluorine Bohr Diagram
Bohr diagram is very interesting and easy to draw. Here, we will draw the Bohr diagram of the Fluorine atom with some simple steps. Steps to draw the Bohr Model of Fluorine atom 1. Find the number of protons, electrons, and neutrons in the Fluorine atom Bohr diagrams show electrons orbiting the nucleus of an atom somewhat like planets orbit around the sun. In the Bohr model, electrons are pictured as traveling in circles at different shells, depending on which element you have. Figure 2 2 contrast the Bohr diagrams for lithium, fluorine and aluminum atoms. The shell closest to the nucleus is. Bohr Diagrams. Bohr diagrams show electrons orbiting the nucleus of an atom somewhat like planets orbit around the sun. In the Bohr model, electrons are pictured as traveling in circles at different shells, depending on which element you have. Figure \(\PageIndex{2}\) contrast the Bohr diagrams for lithium, fluorine and aluminum atoms. Bohr diagrams for hydrogen, helium, lithium, carbon, fluorine, neon, sodium, silicon, chlorine, and argon. Bohr diagrams indicate how many electrons fill each principal shell. Group 18 elements (helium, neon, and argon are shown in Figure 2) have a full outer, or valence, shell. A full valence shell is the most stable electron configuration.
Fluorine chemical elements
For more information and resources, please visit: https://sites.google.com/tdsb.on.ca/htc-meyer/home/9-Science/9-chemistry December 14, 2023 by Deep The information on this page is fact-checked. Fluorine Bohr model In the fluorine Bohr model, the nucleus holds 9 protons and 10 neutrons. Encircling this nucleus are two electron shells, carrying a total of 9 electrons. To draw the fluorine Bohr model, note the 9 protons, 10 neutrons, and 9 electrons. The electron configuration and the orbital diagram are: Following hydrogen is the noble gas helium, which has an atomic number of 2. The helium atom contains two protons and two electrons. The first electron has the same four quantum numbers as the hydrogen atom electron ( n = 1, l = 0, ml = 0, ms = +12 m s = + 1 2 ). By convention, elements are organized in the periodic table, a structure that captures important patterns in their behavior.Devised by Russian chemist Dmitri Mendeleev (1834-1907) in 1869, the table places elements into columns—groups—and rows—periods—that share certain properties.These properties determine an element's physical state at room temperature—gas, solid, or liquid.

Fluorine Electron Dot Diagram
Podcasts Produced by The Naked Scientists. Periodic Table of Videos Created by video journalist Brady Haran working with chemists at The University of Nottingham. Element Fluorine (F), Group 17, Atomic Number 9, p-block, Mass 18.998. Sources, facts, uses, scarcity (SRI), podcasts, alchemical symbols, videos and images. This is the Bohr-Rutherford Diagram for Fluorine (Atomic Number 9). Fluorine is the ninth element of the Periodic Table. As shown, Fluorine has nine protons (i.e., 9p) and ten neutrons (i.e., 10n). Thus, its Atomic Mass is 19. Fluorine also has nine total electrons -- two electrons in Orbit 1, seven electrons in Orbit 2.
In order to make a Bohr diagram, you need to know the number of protons, neutrons, and electrons the element has. In this section, we'll show a sample Bohr diagram for hydrogen. H —Hydrogen 1 proton 1 electron 0 neutrons You can see the principles outlined in the section above at work in the Bohr model for the hydrogen atom. Name: Fluorine Symbol: F Atomic Number: 9 Atomic Mass: 18.998404 amu Melting Point:-219.62 °C (53.530006 K, -363.31598 °F) Boiling Point:-188.14 °C (85.01 K, -306.652 °F) Number of Protons/Electrons: 9 Number of Neutrons: 10 Classification: Halogen Crystal Structure: Cubic Density @ 293 K: 1.696 g/cm 3 Color: Greenish Atomic Structure

Fluorine Diagram
On the far left of Figure 3.6.1 3.6. 1 are the highest energy electromagnetic waves. These are called gamma rays and can be quite dangerous, in large numbers, to living systems. The next lower energy form of electromagnetic waves are called x-rays. Most of you are familiar with the penetration abilities of these waves. Key points Bohr's model of hydrogen is based on the nonclassical assumption that electrons travel in specific shells, or orbits, around the nucleus. Bohr's model calculated the following energies for an electron in the shell, n : E ( n) = − 1 n 2 ⋅ 13.6 eV



